A Single Injection Restored Their Hearing — So Why Aren't Deaf Communities Celebrating?
Summary
An AAV-OTOF gene therapy trial restored hearing in all ten patients with congenital deafness, improving average thresholds from 106 dB to 52 dB, yet the breakthrough has ignited fierce opposition from Deaf communities who view it as an existential threat to their culture and identity. With FDA approval of Regeneron's DB-OTO imminent and China already leading the world with 21 treated patients, this medical milestone forces an uncomfortable reckoning: the collision between a genuine cure and the rights of a linguistic minority, compounded by deep inequities in global access and unresolved ethical questions about treating infants who cannot consent to irreversible changes in their biology.
Key Points
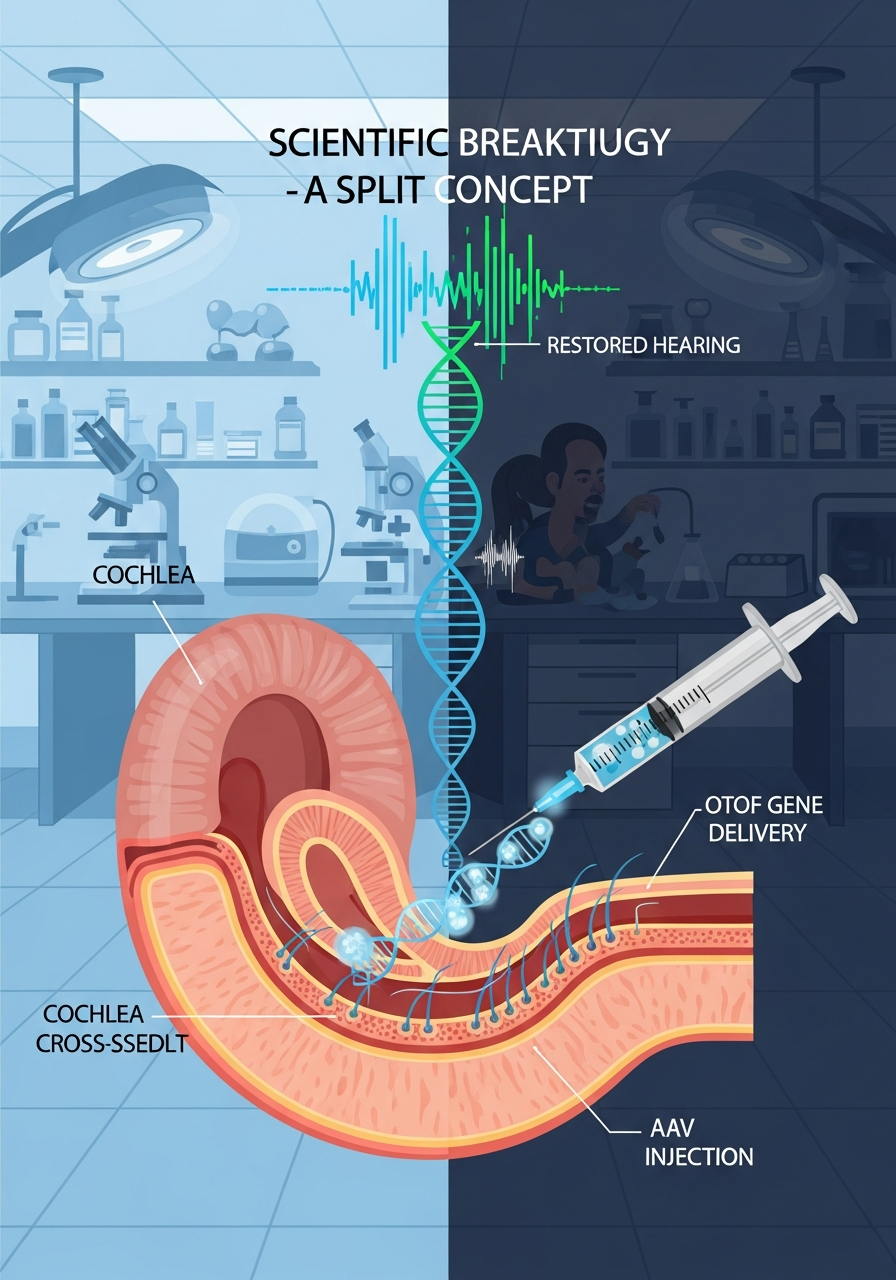
OTOF Gene Therapy Achieves 100% Success Rate Across All Ten Patients
In a joint clinical trial conducted by the Karolinska Institute and five Chinese hospitals, AAV-OTOF gene therapy successfully improved hearing in all ten patients aged 1 to 24. The average hearing threshold dropped from 106 dB to 52 dB, representing a 54 dB improvement.
Deaf Culture vs. the Medical Paradigm — Understanding the Cultural Genocide Debate
The British Deaf Association issued an official statement declaring that deaf children are not broken, while Deaf activists have compared gene therapy to becoming an endangered species.
China Seizes the Lead with 21 Patients Treated — More Than Any Other Country
The world's first gene therapy for hereditary hearing loss was performed at Fudan University in Shanghai in 2022, and Chinese research teams now hold the global record with 21 patients treated.
FDA Approval Imminent — The First Commercially Available Gene Therapy for Hereditary Deafness
Regeneron's DB-OTO has received Orphan Drug, Rare Pediatric Disease, Fast Track, and RMAT designations, and its selection for the FDA's CNPV program has compressed the review period from 12 months to 1 to 2 months.
The Access Divide — Children in Wealthy Nations Will Hear, Children in Poor Nations Will Not
Gene therapy development costs average approximately $1.94 billion, and some treatments carry price tags exceeding $4 million for a single dose.
Positive & Negative Analysis
Positive Aspects
- Overwhelming Therapeutic Efficacy and Safety Profile
A 100% success rate across all ten patients, an average hearing improvement of 54 dB, onset of effects within one month, and no serious adverse events over 6 to 12 months of follow-up.
- Scalability to Other Forms of Hereditary Deafness
While OTOF mutations account for only 2 to 8 percent of hereditary hearing loss, GJB2 mutations are responsible for roughly 50 percent. If GJB2 trials succeed, the treatable patient population expands more than tenfold.
- Expedited FDA Pathway and Regulatory Precedent
DB-OTO accumulation of Orphan Drug, Fast Track, RMAT, and CNPV designations compressing FDA review to 1 to 2 months sets a powerful precedent.
- Long-Term Cost Efficiency Compared to Cochlear Implants
Bilateral cochlear implant surgery costs approximately $100,000 to $150,000, with lifetime expenses adding substantially. Gene therapy addresses the root cause in a single administration.
- International Expert Consensus Establishing a Standardization Foundation
The international consensus reached by 46 multidisciplinary experts produced 30 position statements representing the field's first systematic guidelines.
Concerns
- An Existential Threat to Deaf Culture Itself
If gene therapy becomes standard practice, the number of children growing up Deaf could plummet. Sign languages, Deaf art, Deaf schools face the risk of disappearing within a single generation.
- Fundamental Uncertainty About Long-Term Safety
A follow-up period of 6 to 12 months is woefully insufficient for assessing the long-term effects of gene therapy.
- Deepening Global Access Inequality
With average gene therapy development costs of $1.94 billion and some treatments priced above $4 million per dose, access for patients in developing nations is effectively impossible.
- The Ethical Dilemma of Treating Infants — Identity Decided Without Consent
Data showing optimal outcomes in children aged 5 to 8 creates pressure to treat as early as possible, stripping the child of the opportunity to choose their own identity.
- Regulatory Gaps Creating Uneven Distribution of Safety Risk
China's world-leading record of 21 treated patients is a product of its more flexible regulatory environment, but this flexibility may come at the cost of less rigorous safety verification.
Outlook
The most consequential event on the immediate horizon is the FDA's decision on Regeneron's DB-OTO. With the regulatory submission completed by late 2025 and the CNPV program compressing the review period to just 1 to 2 months, a final approval decision is expected as early as the first half of 2026.
Sources / References
- AAV Gene Therapy for Autosomal Recessive Deafness 9: A Single-Arm Trial — Nature Medicine
- DB-OTO Gene Therapy for Inherited Deafness — New England Journal of Medicine
- Gene Therapy: Who Should Decide the Deaf Community's Future — The Lancet
- British Deaf Association Responds to Gene Therapy Trial to Treat Deafness — British Deaf Association
- International Expert Consensus on Gene Therapy for Hereditary Hearing Loss — ScienceDirect
- Gene Therapies for Deafness Dredge Up an Old Question: Do Deaf People Want a Cure? — STAT News
- Deafness Reversed: One Injection Restores Hearing in Just Weeks — ScienceDaily
- FDA Expediting Review of Regeneron Drug to Treat Genetic Hearing Loss — The Hearing Review
- Gene Therapy Restores Hearing in Deaf Children — Karolinska Institutet Study — Karolinska Institutet
- Regeneron Announces Positive Data from DB-OTO Gene Therapy — Regeneron Investor Relations
- OTOF Gene Therapy: From Breakthroughs to Roadmaps — PMC / MedComm
- For the Deaf Community, Gene Therapy Raises Profound Questions About Identity and Culture — Science (AAAS)
- Eli Lilly / Akouos Reports AK-OTOF Phase 1/2 Clinical Results — Eli Lilly / Akouos