50 Years of Brain Research vs. One Bacterial Sugar Molecule — Who Actually Holds the Key to ALS?
Summary
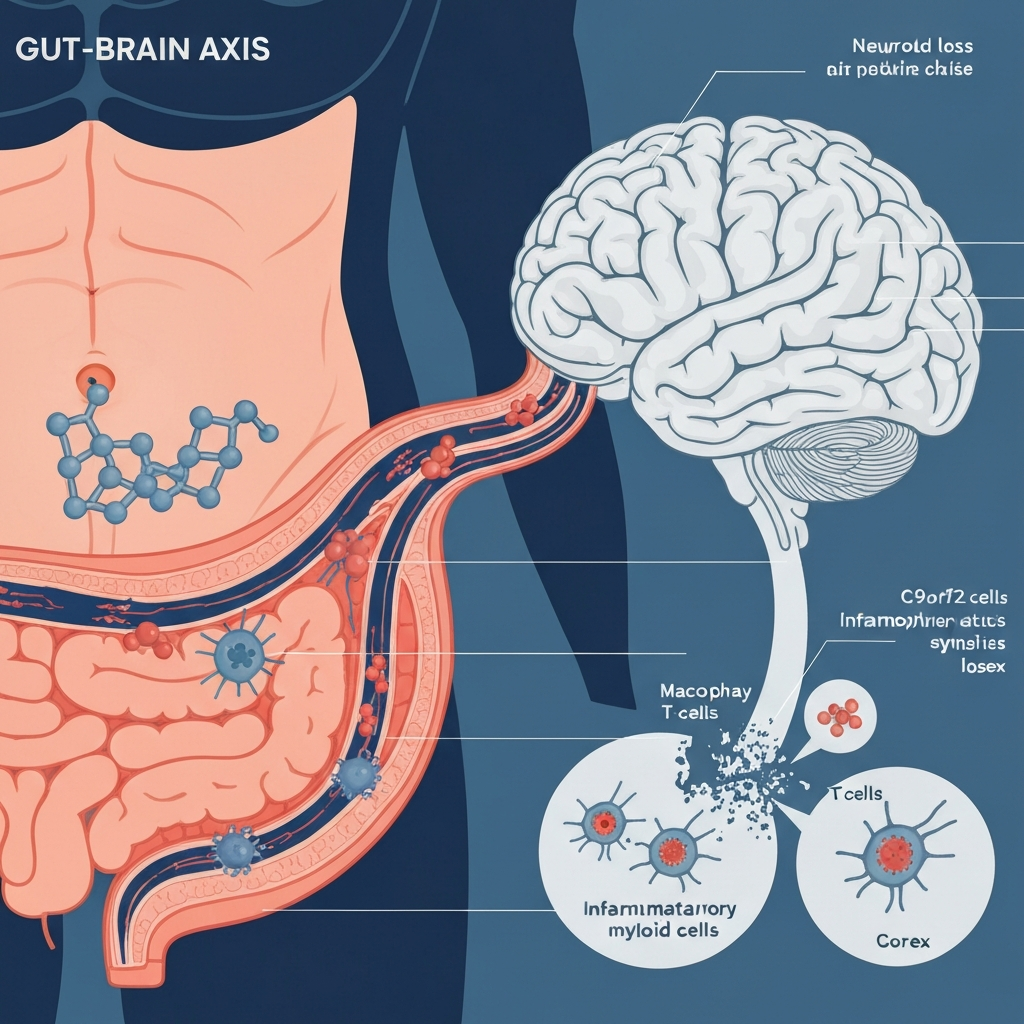
A landmark study published in Cell Reports in April 2026 by Aaron Burberry's research team at Case Western Reserve University presents decisive evidence that the environmental trigger for ALS and frontotemporal dementia (FTD) may originate not in the brain, but in the gut microbiome. Among 23 ALS and FTD patients examined, 70% showed elevated concentrations of bacterial glycogen — an inflammatory sugar molecule produced by intestinal bacteria — compared to only 33% in a healthy control group, a more than twofold difference that fundamentally reframes our understanding of neurodegenerative disease onset. Bacterial glycogen appears to hyperactivate the immune system and drive neuroinflammation, potentially explaining why individuals carrying the same C9orf72 genetic mutation experience wildly divergent fates, with family-based penetrance estimates ranging from 16% to over 60%. Mouse experiments demonstrated that reducing bacterial glycogen improved brain health and extended lifespan, providing early functional evidence of causation rather than mere association. This discovery challenges the brain-centric paradigm that has dominated neuroscience for half a century, while simultaneously demanding honest scrutiny of its own structural limitations: a sample of just 23 patients, and a microbiome research landscape where only 15% of studies employ designs capable of supporting causal inference.
Key Points
70% of ALS and FTD Patients Showed Elevated Bacterial Glycogen — Evidence That Gut Bacteria May Be the Environmental Trigger
The single most striking number in the Cell Reports paper is this: 70% of ALS and FTD patients in the study had elevated concentrations of bacterial glycogen, compared to only 33% in the healthy control group — a more than twofold difference that demands a serious rethinking of where ALS originates. Bacterial glycogen is a polysaccharide that gut bacteria synthesize as an energy storage molecule. Normally it stays inside the intestines, but when the gut barrier becomes compromised — a condition increasingly documented in ALS patients — bacterial glycogen enters the bloodstream, where it encounters immune cells primed to respond aggressively to foreign microbial material. In C9orf72-deficient myeloid cells, that response doesn't get properly regulated, and the result is a cascade of inflammatory cytokines that drives neuroinflammation at a distance from the gut itself. What makes this finding particularly compelling is that Burberry's team didn't just measure a correlation with one bacterial species. They tested ten phylogenetically distinct bacterial strains and found that all ten triggered cytokine explosions in a C9orf72-dependent manner. Metatranscriptomic analysis then identified the glycogen biosynthesis pathway as the unifying inflammatory driver across all of them — pointing toward a pathway-level disease mechanism rather than a quirk of a single microbe. That distinction matters enormously for therapeutic strategy, because targeting a shared pathway is both more tractable and more broadly applicable than targeting any single bacterial species. In my view, this is the mechanistic heart of the entire research arc.
The C9orf72 Gene and Gut Bacteria: Why Identical Mutations Lead to Completely Different Fates
The C9orf72 repeat expansion is the most common known genetic cause of both ALS and frontotemporal dementia, but it is also one of the most perplexing. Carriers of the same mutation face a 50% cumulative disease probability by age 58, rising to 99.5% by age 83 — yet indirect family-based estimates put penetrance at just 24–33%, with family-to-family variation ranging from 16% to 60.6%. Siblings with identical genomes can have utterly different lives: one develops ALS in their fifties, the other reaches their eighties symptom-free. The brain-centric model had no satisfying answer for this variability. Protein aggregation, mitochondrial dysfunction, and oxidative stress don't explain why the same gene behaves so differently in the same family across different environments. The gut microbiome, however, provides a genuinely plausible and testable explanation. Environmental variables — diet, antibiotic exposure, geographic location, early-life microbial colonization patterns — shape gut microbiome composition in ways invisible to genetic testing but potentially decisive for disease expression. The 2020 Nature study is the clearest evidence: genetically identical C9orf72-deficient mice from two research facilities had dramatically different survival outcomes, explained entirely by differences in their gut microbiome compositions. The Harvard mice died early; the Broad Institute mice lived normally. The mortality gap was statistically significant at P=0.0179. When gut bacteria alone determine whether genetically identical animals live or die, you have crossed the line from correlation into something that begins to look structurally like causation.
Reducing Bacterial Glycogen Extended Lifespan in Mouse Models — Functional Evidence of Causation
The scientific community is rightly cautious about inferring causation from microbiome research, and for good reason — the field has been plagued by correlational studies that collapse under scrutiny. What distinguishes this research from the typical microbiome paper is the presence of functional intervention data. Burberry's team didn't just observe that bacterial glycogen levels were higher in sick patients. They manipulated bacterial glycogen concentrations and measured the outcomes. When glycogen was reduced, brain health measurably improved and lifespan extended. This shifts the claim from "bacterial glycogen is associated with ALS" to "bacterial glycogen reduction improves ALS-like outcomes" — a fundamentally different and much stronger scientific statement. The 2020 Nature study adds another layer: broad-spectrum antibiotic treatment cleared the gut microbiome and reduced inflammation in C9orf72-deficient mice, while fecal microbiota transplant from healthy environments diminished the disease phenotype. Taken together, these results satisfy what scientists call a "functional causal" criterion at the preclinical level — the intervention changes the outcome in the predicted direction. Does that guarantee translation to humans? No — human and murine gut ecosystems are fundamentally different, and many promising mouse findings have failed in human trials. But it does mean this is no longer a purely observational story, and that distinction is what gives the research team scientific standing to propose human trials. Among the 2,397 ALS-microbiome studies reviewed in the 2026 Frontiers systematic review, only 61 met basic methodological standards. This research is among the strongest of that 2.5% in terms of mechanistic depth.
The Microbiome Therapeutics Market Is Exploding — and This Study Just Added Fuel
The commercial implications of this research sit on top of market trends that were already moving fast before this paper landed. Precedence Research projects the microbiome therapeutics market will grow from $250 million in 2025 to $3.4 billion by 2034, at a CAGR of 33.67%. The broader human microbiome market — spanning diagnostics, therapeutics, and consumer health — is projected to leap from $1.23 billion in 2025 to $18.27 billion by 2035 at a CAGR of 30.97%. These are extraordinary growth rates, and the Burberry study adds a high-profile neurological indication to a market previously dominated by gastrointestinal diseases. The ALS treatment market itself sits at $667 million as of 2023, projected to reach $988 million by 2030 at 5.8% CAGR — projections that predate any credible gut-brain axis therapeutic candidate entering the space. What makes this commercially significant beyond the raw numbers is the technology readiness of the platforms involved. FMT is already FDA-approved for a separate indication, with years of safety data. Targeted probiotics and synbiotics have established manufacturing infrastructure. The path from this paper to a clinical product is meaningfully shorter than it would be if the underlying technology were completely novel. I believe we are watching the early formation of a cross-indication microbiome therapeutics platform, where ALS and FTD serve as high-profile anchor indications that attract the research investment and regulatory attention to mature the field broadly — raising the commercial ceiling for the entire space.
The 23-Patient Limitation — Why the Most Important Number in This Study Is Also Its Biggest Weakness
Here's something the press coverage will almost universally undersell: the 70% figure that everyone is citing comes from 23 patients. The healthy control group is even smaller. This is not a fatal flaw in the study's scientific contribution — hypothesis-generating research doesn't require large cohorts, and the mechanistic work in this paper is genuinely rigorous. But it is a critical filter that should change how this evidence is used and communicated publicly. A 2025 consensus statement in Nature Reviews Gastroenterology found that across the microbiome research literature, only 15% of studies employ designs capable of supporting causal inference; 30% fail to adequately control for confounding variables; and over 40% don't report clinically meaningful effect sizes. The 2026 Frontiers systematic review of ALS-microbiome research found that of 2,397 published studies, only 61 — 2.5% — met basic methodological standards. The Burberry study is one of the better-designed pieces in a weakly designed field. But "stronger than average in a weak field" is not the same as "ready for clinical translation." The reverse causation problem remains live: ALS patients experience dysphagia, dietary changes, reduced activity, and autonomic shifts that can alter gut microbiome composition as consequences of the disease rather than causes. What would truly settle the causation question is a longitudinal cohort of pre-symptomatic C9orf72 carriers followed for a decade or more — a study that does not yet exist and won't yield meaningful data for at least 10 years. I believe the intellectually honest framing is: this is the most compelling current evidence for a gut-brain connection in ALS, and it demands urgent, rigorous, and transparently skeptical follow-up.
Positive & Negative Analysis
Positive Aspects
- The Blood-Brain Barrier Problem Disappears Entirely
For the past two decades, the single biggest engineering obstacle in ALS and Alzheimer's drug development has been the blood-brain barrier. Large-molecule therapeutics — antibodies, mRNA-based drugs, gene therapies — lose much of their efficacy before ever reaching neural tissue. The delivery problem has consumed billions in R&D and delayed countless promising candidates by years, creating an entire sub-industry of drug delivery engineering with limited success in truly refractory CNS conditions. If the therapeutic target shifts to the gut, that problem simply ceases to exist in its current form. Drugs and interventions acting on the intestinal microbiome operate within the gastrointestinal tract, where the barrier challenge is categorically different and dramatically lower. Fecal microbiota transplants, targeted probiotics, synbiotics, dietary fiber interventions, and bacteriophages that selectively suppress glycogen-producing bacterial strains all operate in a therapeutic space that is already well-characterized and commercially established. The manufacturing infrastructure exists, the regulatory pathways are established, and the clinical trial expertise is transferable from existing GI indications. In my view, this single feature — circumventing the BBB entirely — makes gut-brain axis approaches one of the most economically and scientifically attractive areas in neurodegenerative medicine for the coming decade. Combination regimens pairing a gut intervention with an existing brain-targeted drug like riluzole or edaravone could deliver synergistic effects without requiring any new CNS drug delivery innovation at all.
- An Early Warning System for ALS Finally Becomes Conceivable
One of the most heartbreaking aspects of ALS is that by the time of diagnosis, patients have typically already lost 50–80% of their motor neurons. The disease is devastating partly because it cannot be caught early enough to intervene meaningfully. Bacterial glycogen could change that calculus in a fundamental way. If bacterial glycogen concentrations in stool or blood correlate with disease risk before symptoms emerge — and the current evidence is consistent with that hypothesis — then for the first time in ALS's history, we have a candidate presymptomatic biomarker that is measurable, accessible, and potentially modifiable. C9orf72 carriers could begin regular bacterial glycogen monitoring in their late thirties or forties, with a defined threshold that triggers clinical intervention: a microbiome rebalancing protocol, a dietary change, a targeted probiotic, or an escalation in monitoring intensity. The penetrance variability that has puzzled clinicians for decades — 16% to 60.6% across families — would finally have a testable environmental correlate, transforming a probabilistic anxiety into a manageable surveillance program. I expect that within 18 months, commercial microbiome testing platforms will have launched some version of a "neurodegeneration risk panel" featuring bacterial glycogen as a marquee marker — initially without robust clinical validation, but creating the market pressure that drives academic validation studies to follow rapidly. This is how biomarker categories get established in practice, and it is happening faster than most clinicians currently anticipate.
- Existing Approved Drugs Get a Second Chance Through Combination Therapy
Riluzole extends ALS survival by approximately two to three months. Edaravone slows functional decline in a defined subset of patients. Tofersen reduces SOD1 protein levels in a specific genetic cohort. These are real but limited benefits — and they have been limited for decades, because every approved ALS drug targets a single pathway in an exclusively brain-centric model. The introduction of gut-brain axis therapy doesn't make these drugs obsolete. It potentially makes them substantially more effective through combination. Here's the biological logic: if gut-derived bacterial glycogen is the upstream driver of the neuroinflammatory environment that accelerates motor neuron death, and you reduce that inflammatory burden through microbiome intervention, then downstream drugs like riluzole — operating on glutamate toxicity — may be working in a less hostile neural environment, with effects that are maintained longer and applied against a less advanced disease process. Think of it as clearing a flooded road before trying to drive on it. Combination trial design for these regimens is relatively accessible: you don't need novel drug approval, you need a Phase 2 add-on design comparing standard-of-care alone versus standard-of-care plus gut intervention. That design could be in patient enrollment within 18 months at well-resourced academic medical centers, and it represents the fastest possible route from this research to a meaningful patient benefit in the near term.
- FMT and Proven Microbiome Technologies Are Already Deployable
One of the strongest practical arguments for this research avenue is that it doesn't require building therapeutic capability from scratch. Fecal microbiota transplant was FDA-approved in 2023 for recurrent C. difficile infection, with years of safety data and established clinical protocols at major academic medical centers. The 2025 Frontiers in Nutrition review documented FMT's positive effects on respiratory function and neuromuscular parameters in ALS patients across multiple small studies. Synbiotic formulations — combining targeted probiotics with prebiotic fibers — have demonstrated predictable engraftment and butyrate elevation in controlled studies. Even bacteriophages capable of selectively targeting specific bacterial strains are advancing through regulatory review in the EU and Asia. What this means practically is that the clinical infrastructure for gut microbiome intervention in a neurological context already exists. You don't need a decade of manufacturing development before the first human trial. The Phase 1 safety studies that Burberry's team says are feasible within a year aren't starting from conceptual zero — they're repositioning validated platforms toward a new indication with a mechanistic rationale that is more specific than anything previously available. Sodium Oligomannate's FDA clearance for a Phase 2 Parkinson's trial in 2025 illustrates exactly this dynamic: an established compound with gut microbiome effects entering the neurological indication space because the platform was already proven elsewhere.
- This Could Redefine Neurodegenerative Disease as Preventable, Not Just Treatable
The most profound long-term implication of this research is the possibility of reclassifying ALS and FTD from reactive-treatment conditions to proactive-prevention conditions. Right now, the medical encounter for ALS begins at diagnosis — which means it begins at a point of irreversible neurological damage. If bacterial glycogen monitoring becomes a standard component of care for genetically at-risk individuals starting in middle age, the intervention window shifts from post-damage to pre-damage. That is a category change in medicine, not an incremental improvement. The parallel to Alzheimer's research is instructive: the concept of "biological Alzheimer's disease" — defined by biomarker profiles before cognitive symptoms emerge — has already transformed how clinical trials are designed and how at-risk individuals are counseled and monitored. A similar conceptual shift in ALS, anchored by a gut-derived biomarker that is measurable, accessible, and mechanistically linked to disease expression, would be one of the most significant advances in neurology in a generation. I believe this is the scenario that deserves the most serious investment of research resources right now — not because it's guaranteed, but because the cost of not pursuing it, if the hypothesis proves correct, is measured in lives lost during the years we spent being cautious rather than bold.
Concerns
- A Sample of 23 Is a Hypothesis, Not a Conclusion — and Media Doesn't Know the Difference
The 70% figure will be everywhere. What will be invisible is the denominator: 23 patients. Frontiers' 2026 systematic review of 2,397 ALS-microbiome studies found that only 2.5% met basic methodological standards. Nature Reviews Gastroenterology's 2025 consensus statement found that 30% of microbiome studies fail to control for confounders adequately, and over 40% don't even report clinically meaningful effect sizes. A single Cell Reports paper with 23 patients, however rigorously designed, does not overcome this structural landscape. The media coverage pattern is entirely predictable: the 70% number gets reported without the sample size, without the confidence intervals, without the caveat that this is hypothesis-generating research. Families of ALS patients begin searching for whatever is closest to "bacterial glycogen treatment" online, and supplement companies fill that search vacuum within weeks. I believe researchers and institutions have an ethical obligation to be proactive — explicitly stating in every public communication that this study demonstrates a promising mechanistic hypothesis requiring large-scale replication, not a proven causal relationship ready for clinical adoption. The scientific community's failure to communicate this distinction clearly and consistently has real-world consequences: they play out in the wallets and shattered expectations of the most vulnerable people following this story with desperate hope.
- The Reverse Causation Problem Has Not Been Fully Resolved
The uncomfortable truth about ALS and gut microbiome research is that the causal arrow could point in either direction. ALS patients experience profound systemic changes as the disease progresses: dysphagia alters dietary intake, reduced mobility changes gut motility, respiratory compromise affects the autonomic nervous system that regulates intestinal function, and medication regimens further perturb the microbial environment. All of these consequences of ALS can independently shift gut microbiome composition — and potentially elevate bacterial glycogen concentrations — as effects of the disease rather than causes of it. The 2026 Frontiers review captures this ambiguity precisely: "dysbiosis may contribute to, accompany, or result from neurodegeneration." The mouse data from Nature 2020 provides meaningful directional evidence — gut bacteria differed before full disease expression, and manipulating them changed outcomes — but mouse gut ecology is fundamentally different from human gut ecology in ways that consistently confound translational neuroscience. The Burberry study's combination of human patient data with mouse functional experiments represents the strongest current methodological approach available, but it is not a longitudinal cohort study of pre-symptomatic C9orf72 carriers measured before symptoms emerge, which is what would truly settle the causal question. That study doesn't exist yet, and it won't produce meaningful findings for at least a decade. Until it does, reverse causation remains a live alternative hypothesis.
- This Finding Applies to 5–10% of ALS Patients — What About Everyone Else?
C9orf72 repeat expansions account for 5–10% of all ALS cases. The bacterial glycogen pathway is mechanistically anchored to C9orf72 function in myeloid cells. What does this mean for the other 90–95% of ALS patients who don't carry the C9orf72 mutation — the vast majority of people living with this disease worldwide? The honest answer is: we have no idea. The current evidence provides no mechanistic basis for assuming the bacterial glycogen pathway drives sporadic ALS, which accounts for the overwhelming majority of cases. This is a critical constraint on both the scientific and commercial potential of the discovery. From a drug development economics perspective, a therapeutic that serves only 5–10% of an already rare disease population faces severe commercial headwinds — high development costs amortized across a tiny eligible patient population, with limited pricing power even at orphan drug premiums. From a clinical and ethical perspective, if pharma companies rush to position bacterial glycogen-targeting therapies as general ALS treatments rather than C9orf72-specific interventions, they are setting up Phase 3 failures that will discredit the entire approach for years. The responsible path is clear: be explicit about the genetic scope of current evidence, design initial trials rigorously within that scope, and pursue broader applicability as a separate subsequent scientific question that requires its own evidence base.
- The Amyloid Hypothesis Failure Is a Warning This Field Must Not Ignore
Neuroscience has been here before, and it did not end well. Starting in the 1990s, the amyloid hypothesis — the idea that amyloid beta plaques are the primary driver of Alzheimer's disease — became the dominant paradigm in the field. Hundreds of billions of dollars flowed into amyloid-targeting drug development over thirty years. The vast majority of those drugs failed at Phase 3. The failures weren't random or entirely unpredictable — there were warning signs throughout that the field largely rationalized away in favor of continuing along the established research track. The drugs that eventually reached approval — aducanumab and lecanemab — cleared amyloid successfully but produced only modest cognitive benefits that remain genuinely contested in the clinical community. The amyloid hypothesis wasn't entirely wrong; it just wasn't complete or primary enough to carry the entire therapeutic strategy of a field. The gut-brain axis hypothesis in ALS faces analogous risks. A compelling mechanism at small scale, with strong biological plausibility and enthusiastic early adopters, is exactly the profile of every promising hypothesis before the large-scale trials begin. I believe ALS and FTD researchers need to pursue the bacterial glycogen pathway aggressively while simultaneously maintaining investment in the neuroinflammation, glutamate toxicity, and protein homeostasis pathways that remain mechanistically distinct. Paradigm concentration — betting everything on one mechanism — is precisely the error mode that produced three decades of amyloid disappointment.
- The Supplement Market Will Exploit This Before Regulators Can Respond
The microbiome consumer market is already a regulatory gray zone populated with products making claims that range from optimistic to outright fraudulent. Within weeks of the Cell Reports publication, e-commerce platforms in the US, Europe, and Asia will see proliferating products tagged with terms like "ALS prevention," "brain health probiotic," "gut-brain support," and "neurodegeneration defense." None of these products will have evidence supporting ALS-relevant efficacy. Most will contain bacterial strains with no demonstrated effect on glycogen biosynthesis pathways. Some will contain no meaningful active ingredients at all beyond standard probiotic blends that have never been tested in any ALS-adjacent context. The harm here is twofold: ALS patient families spend money they often can ill afford on products that cannot help them, and every dollar spent on unproven supplements represents a dollar not invested in validated clinical programs that might actually advance the science. The FTC and FDA will eventually respond with warning letters and enforcement actions — but regulatory response consistently lags market behavior by 12–24 months, and even then enforcement is selective rather than comprehensive. The most effective near-term intervention is preemptive: the research community, university communications offices, and patient advocacy organizations like the ALS Association need to issue clear, explicit, publicly prominent guidance stating that no currently available supplement or probiotic product has been validated for ALS prevention or treatment based on this research.
Outlook
Let's start with the near-term picture — the next one to six months — because I think what happens first will surprise most people. The scientific community will move slowly by necessity. Clinical trials don't launch the month a paper is published; patient recruitment alone takes 12–18 months, and IRB approval layers on additional time. But the market will not wait. Google Trends data already shows spikes in "gut bacteria ALS" searches following the Cell Reports publication, and microbiome-adjacent stocks have posted double-digit percentage gains in the days since. Companies like Seres Therapeutics, Finch Therapeutics, and Vedanta Biosciences — already operating in IBD and C. difficile with established microbiome platforms — are almost certainly running internal protocol assessments right now, evaluating whether existing pipeline assets can be repositioned for ALS indications.
The near-term bull scenario is that Burberry's team or a competing lab identifies a specific probiotic candidate or bacteriophage capable of reducing bacterial glycogen concentrations at meaningful scale, triggering a funding rush and a wave of startup launches. The near-term bear scenario is that a competing group publishes a small-scale replication failure, cooling the hype cycle before it fully ignites. My realistic base case lands between these poles: diagnostic tooling moves faster than therapeutics. Consumer-grade microbiome platforms like Viome and Zoe are almost certainly drafting "neurodegeneration risk panel" beta features that include bacterial glycogen as a marquee marker, months before any therapeutic candidate clears Phase 1. That is the shape of early commercial adoption in this space — diagnostics first, drugs years later.
The medium-term window — roughly six months to two years out — is where the scientific and commercial timelines begin visibly diverging. The first major trend is the launch of Phase 1 and Phase 2 clinical trials. The most likely initial cohort is C9orf72 mutation carriers with documented family history — not because all ALS patients share this mechanism, but because C9orf72 is the only genetic anchor we currently have for the bacterial glycogen pathway. These early-phase trials will assess safety, target engagement, and initial efficacy signals: measuring bacterial glycogen concentrations in stool and blood, testing targeted probiotic and FMT protocols, and tracking inflammatory biomarker changes over months. If Phase 2 efficacy signals come in positive, the microbiome therapeutics market — projected at $250 million in 2025 and $3.4 billion by 2034 per Precedence Research — will see meaningful upward revision in ALS-specific allocation. The ALS treatment market, at $667 million in 2023 and projected at $988 million by 2030 at a 5.8% CAGR per Grand View Research, could tip into double-digit growth if gut-brain therapies deliver even modest efficacy data.
The second medium-term trend is biomarker commercialization racing ahead of therapeutics. The regulatory question of whether bacterial glycogen assays get classified as laboratory-developed tests (LDTs) or regulated as in vitro diagnostic devices (IVDs) will become a live battleground in the US, the EU, and across Asia. The EU's IVDR framework would almost certainly push these assays toward full IVD classification. South Korea's amended In Vitro Diagnostic Medical Device Act is already moving faster than most observers expected, and Japan's regulatory posture is similarly tightening. My expectation is that by the 18-month mark, at least two or three companies will have commercial bacterial glycogen panels available — primarily positioned as "family history risk stratification" tools for C9orf72 carriers. This is the "slow drug, fast diagnostic" trajectory I find most plausible as the medium-term base case.
The third medium-term trend is the restructuring of the ALS treatment landscape itself. Right now, approved ALS therapies — riluzole, edaravone, tofersen — extend survival by months, not years. These are brain-centric, single-pathway interventions. The introduction of gut-brain axis therapies opens the door to combination protocols working through orthogonal mechanisms. If you lower baseline gut-driven neuroinflammation first and then apply riluzole's glutamate toxicity suppression on top, the synergy could substantially amplify efficacy. The medium-term bear scenario is that early human FMT studies fail to replicate mouse findings at P<0.05, or reveal significant adverse events from broad-spectrum antibiotic co-treatment, stalling the pipeline. My base case is that therapeutics grind carefully through Phase 1 and 2 while diagnostics enter commercial markets aggressively — and the companies that design combination trial protocols earliest hold a durable first-mover advantage.
Looking out two to five years, I think the probability of this research reshaping the paradigm for neurodegenerative disease broadly sits at better than 50%. The single most transformative possibility is the redefinition of ALS and FTD as presymptomatic diseases — conditions detectable, monitored, and intervened upon before the first motor neuron dies. This is not a subtle conceptual shift. It is the difference between ALS as a death sentence handed down after symptoms emerge, and ALS as a manageable chronic risk factor addressed proactively starting in your forties. If bacterial glycogen monitoring becomes standard care for C9orf72 carriers beginning at age 40, with targeted microbiome interventions triggered at defined threshold values, we would be doing something medicine has never been able to do for ALS: genuine prevention. The long-term bull scenario has Phase 3 delivering significant efficacy data, FDA granting conditional approval for a bacterial glycogen-targeting therapeutic, and the gut-brain axis framework cascading from ALS and FTD into Parkinson's and Alzheimer's — a platform shift that makes this one of the great inflection points in modern neuroscience.
But the bear scenario deserves equal weight. The amyloid hypothesis in Alzheimer's disease is the cautionary tale this field must never forget. For thirty-plus years, the neuroscience establishment channeled trillions into amyloid-targeting drugs. Most failed catastrophically at Phase 3. The structural problem is that promising mechanisms in small studies frequently collapse when scaled. The causal inference deficit in microbiome research — only 15% of studies capable of supporting causal inference — is not a footnote. It means that when bacterial glycogen moves from 23 patients to a 500-person randomized controlled trial, we have no guarantee the association holds at clinical scale. My realistic base case sits deliberately between extremes: Phase 1 and 2 deliver strong signals, Phase 3 produces partial efficacy earning conditional approval specifically for C9orf72-variant ALS, and bacterial glycogen therapy becomes a precision medicine option for a defined genetic subset while the remaining 90–95% of ALS patients still await their answer. I place the probability of the bull scenario at roughly 55% and the bear at 45% — but either way, neuroscience cannot return to "brain only" as an organizing premise.
What does all of this mean for different groups watching this story unfold? For individuals with C9orf72 family history: nothing is clinically actionable today, but within one to two years, clinically meaningful bacterial glycogen panels are likely to exist. For general readers: the clearest danger is the supplement market. No currently available probiotic has been validated for ALS or FTD prevention based on this research — do not let marketing outpace science. For investors: diversified exposure to the microbiome diagnostics and therapeutics platform is smarter than concentrated bets on bacterial glycogen alone; companies with FMT infrastructure and regulatory track records are better positioned than speculative startups. For policymakers in rapidly aging economies — South Korea, Japan, China — the gut-brain axis represents a domestically accessible frontier where existing probiotic manufacturing and FMT infrastructure can be mobilized faster than waiting for imported CNS drugs.
Here's the question I keep coming back to, and I think it's the most important thing this paper is really asking: what else are we refusing to look at right now? The evidence linking gut health to brain disease didn't appear from nowhere in 2026. The Nature 2020 paper existed. Reviews from 2015, pilot data from 2010 — the signal has been quietly accumulating. Yet clinical pipelines remained uniformly brain-centric. That gap is not a scientific failure. It is a deeply human institutional failure — we look for answers where we already know how to look, where funding already flows, where career incentives already point. The lesson here isn't only about bacterial glycogen. It's about whether science can build institutions willing to look where the map currently says "here be dragons." If we want the next 50 years of neuroscience to be more productive than the last 50, this ALS discovery needs to be more than a single paradigm-challenging paper. It needs to be the trigger for systematic rethinking of how we fund, design, and evaluate research at the boundaries between neurology, immunology, and microbiology.
One thing needs to be stated plainly before we go further: this article is an analytical essay about a research hypothesis at the academic stage. No probiotic, FMT protocol, or dietary intervention targeting bacterial glycogen has received regulatory approval for ALS or FTD prevention or treatment as of April 2026. If you or someone in your family carries C9orf72 or has ALS symptoms, please consult a neurologist and a genetic counselor before making any decisions about supplements or microbiome interventions. Every individual treatment decision should be made with a qualified clinician — not based on a single research paper, however promising, and not based on this essay. The goal here is to analyze where the science is heading, not to prescribe a course of action.
Five years from now, if we are calling ALS a "gut-immune-brain network disease" rather than simply a "brain disease," the April 2026 Cell Reports paper will be recorded as the inflection point. And if that shift doesn't materialize — if Phase 3 fails, if the hypothesis collapses under scale — we will at minimum have closed the chapter on the era when neuroscience could claim the brain was the only organ worth studying. Either way, the map has already changed. The key was in the gut all along, and we simply never thought to look there until a sugar molecule 23 patients happened to share forced the question.
Sources / References
- C9orf72 in Myeloid Cells Prevents an Inflammatory Response to Microbial Glycogen — ScienceDaily / Cell Reports
- C9orf72 Suppresses Systemic and Neural Inflammation Induced by Gut Bacteria — Nature / PubMed Central
- Gut Microbiota and ALS: Cause, Consequence or Correlation? — A Systematic Review — Frontiers in Neuroscience
- Age-Related Penetrance of the C9orf72 Repeat Expansion — Scientific Reports / Nature
- Microbiota–Gut–Brain Axis in Neurodegenerative Diseases: Molecular Mechanisms and Therapeutic Targets — Molecular Biomedicine / PubMed Central
- A Consensus Statement on Establishing Causality, Therapeutic Applications and the Use of Preclinical Models in Microbiome Research — Nature Reviews Gastroenterology & Hepatology
- ALS Treatment Market Analysis Report 2024–2030 — Grand View Research
- Microbiome Therapeutics Market Forecast 2025–2034 — Precedence Research
- ALS Prevalence Projected to Rise Sharply Worldwide by 2040 — ALS News Today
- Gut Microbes: The Difference Between Life and Death in C9ORF72 Mice — ALZFORUM
- FDA-Approved ALS Drug Relyvrio Fails Phase III, to Be Withdrawn — Chemical & Engineering News (C&EN)