The World's First Lab-Grown Oesophagus Just Worked in Pigs — And It Could Change Everything About Organ Medicine
Summary
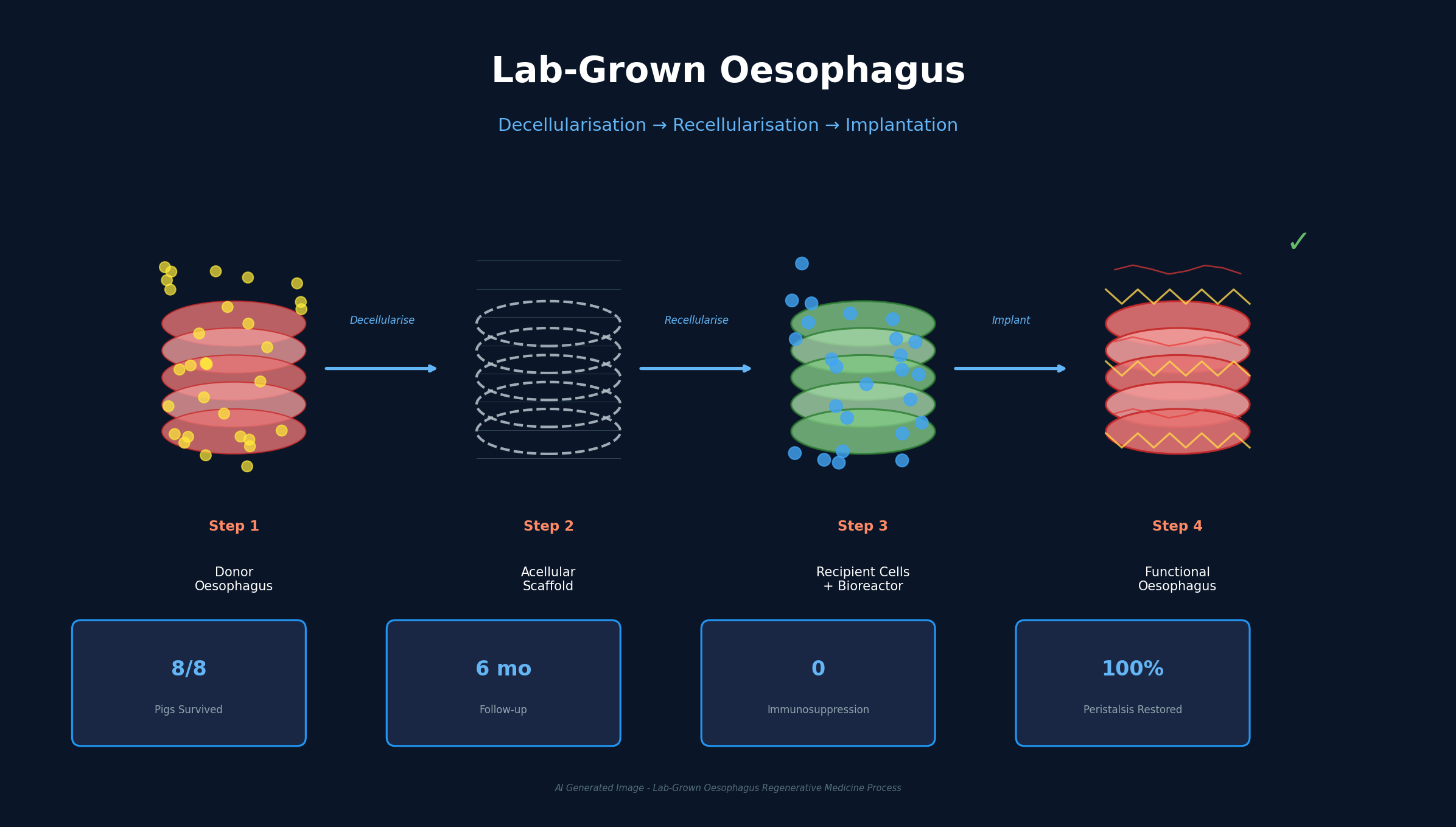
A donor pig oesophagus was stripped of cells, repopulated with the recipient's own cells, and implanted without immunosuppression — restoring normal swallowing for six months. Children born with oesophageal atresia may soon have a fundamentally different treatment option.
Key Points
World-first complete decellularisation-recellularisation pipeline
UCL and GOSH scientists chemically removed all cells from a donor pig oesophagus to create a natural scaffold preserving the organ's 3D architecture. They then harvested muscle cells from the recipient pig via small biopsy, expanded them massively in the lab, and injected them into the scaffold. The seeded scaffold spent one week in a bioreactor circulating growth fluids, with the entire manufacturing process taking approximately two months. Previous studies achieved partial success at various stages, but this is the first time the full pipeline from decellularisation through implantation to functional recovery has been completed.
Six months of full functional recovery without immunosuppression
All eight pigs survived the first month post-implantation, and by six months had developed functioning muscle layers, nerve networks, and blood vessels — all without a single immunosuppressive drug. Full peristalsis was restored, enabling normal food transport to the stomach. In conventional organ transplantation, lifelong immunosuppression is mandatory, increasing infection vulnerability and cancer risk by 2-4x. This approach fundamentally eliminates that requirement by using the recipient's own cells, so the immune system recognizes the organ as self.
Growing animal model validates pediatric application potential
The experiments were conducted in young, rapidly growing pigs whose internal organs must keep pace with body growth. The fact that implanted oesophagi adapted to this growth provides strong evidence for applicability in pediatric patients — the exact population that needs this technology most. Oesophageal atresia affects 2.3-2.6 per 10,000 births globally, with current treatment relying on complex reconstructive surgery that sacrifices healthy stomach or colon tissue as a substitute.
Platform technology extensible to all tubular organs
The decellularisation-recellularisation approach is not limited to the oesophagus. It is a foundational technology applicable across tubular organs including the trachea, small intestine, bladder, and blood vessels. Professor Paolo De Coppi noted this could pave the way for translation to other disease areas. With over 100,000 people on organ transplant waiting lists worldwide and 17 dying daily in the US alone waiting for organs, this platform technology's potential impact extends far beyond a single rare pediatric condition.
$43.1 billion tissue engineering market provides commercial runway
The tissue engineering market, valued at approximately $19.3 billion in 2024, is projected to reach $43.1 billion by 2030 at a 14.3% CAGR. Simultaneous advances in biomaterials, 3D bioprinting, stem cell research, and gene editing are driving expansion. The Asia-Pacific region is forecast to grow fastest at over 19% annually. The UCL-GOSH breakthrough demonstrates not just academic significance but commercial viability, likely attracting investment from biotech firms including Organogenesis, United Therapeutics, and Miromatrix Medical.
Positive & Negative Analysis
Positive Aspects
- Provides a fundamental treatment for children with oesophageal atresia without sacrificing healthy organs
Current long-gap oesophageal atresia treatment relies on complex reconstructive surgery that damages healthy stomach or colon tissue. Lab-grown oesophagi could provide complete replacement without that sacrifice. All eight pigs surviving with normal swallowing function restored over six months validates both safety and efficacy at the preclinical level.
- Elimination of immunosuppression revolutionizes pediatric transplant outcomes
Current organ transplant recipients must take immunosuppressive drugs for life, increasing infection susceptibility and raising cancer risk 2-4x. For pediatric patients facing decades of these medications, the quality-of-life impact is severe. The decellularisation-recellularisation approach eliminates this burden entirely by using the child's own cells, removing an entire category of post-transplant morbidity.
- $43.1 billion tissue engineering market growth provides commercial tailwinds
Growing from $19.3 billion in 2024 to a projected $43.1 billion by 2030 at 14.3% CAGR, the tissue engineering market is powered by simultaneous advances in biomaterials, 3D bioprinting, and stem cell research. Asia-Pacific leads growth at 19%+ annually. This research demonstrates commercial viability beyond academic significance.
- Platform technology scalable across multiple tubular organ types
The approach works as a foundational technology for trachea, small intestine, bladder, and blood vessels — not just the oesophagus. One proven success creates a template for multiple disease areas. With thousands dying annually on organ transplant waiting lists, the ripple effect potential is enormous.
- Nature Biotechnology publication ensures academic credibility and reproducibility
Publication in a top-tier peer-reviewed journal validates methodology and results. Backed by GOSH Charity and UK MRC funding, the research has a sustainable foundation for follow-up studies. Independent replication attempts by regenerative medicine groups worldwide will follow.
Concerns
- Animal model success does not guarantee human clinical translation
Despite anatomical similarity between pig and human oesophagi, subtle differences in immune response, human cell proliferation characteristics, and long-term complication profiles can only be assessed through actual human trials. Medical history is filled with dramatic animal successes that failed to translate — from gene therapies to artificial hearts.
- Two-month manufacturing timeline incompatible with neonatal emergencies
Oesophageal atresia is diagnosed at birth as an emergency, and neonates cannot survive without oesophageal function for two months. The technology requires either prenatal diagnosis for advance preparation or a staged approach with conventional emergency surgery first and bioengineered replacement later. Either scenario demands substantial clinical protocol changes and additional regulatory approval.
- High initial costs and developed-country concentration risk deepening healthcare inequality
Custom organ manufacturing involves expensive steps: cell harvesting, culturing, bioreactor operation, quality assurance, and surgical implantation. Initially available only at elite medical institutions in the UK, US, and Japan. Patients in developing countries with the greatest need could remain excluded for years. Market growth reflects expansion, not democratization of access.
- Unclear regulatory frameworks could delay approval by 10+ years
Lab-grown organs occupy a novel category — neither drugs nor medical devices. FDA CBER, EMA, and MHRA have not fully established clear approval frameworks for living therapeutics. The regulatory pathway for cell therapy and tissue engineering products is considerably more complex than for conventional pharmaceuticals. Expert projections of 3-5 years to first trials and 10+ years to approval are realistic.
- Six-month follow-up insufficient to guarantee decades of durability
While six months demonstrates functional recovery, it is inadequate for guaranteeing decades of durability. Whether implanted oesophagi function normally at 10-20 years, whether unexpected tissue degeneration or tumor risk emerges — these questions remain unanswered. For pediatric patients who must live with these organs through growth and aging, long-term safety data is critically important.
Outlook
In the near term, over the next one to six months, the most visible impact will be an explosion of academic and industry attention. Having been published in Nature Biotechnology, the protocol will be scrutinized and replicated by regenerative medicine labs worldwide. The UCL-GOSH team, already backed by the Medical Research Council and GOSH Charity, will almost certainly secure additional research funding. On the industry side, licensing inquiries and co-development proposals from biotech companies like Organogenesis, United Therapeutics, and Miromatrix Medical are likely. Animal model data will be supplemented with 12-month and 24-month follow-up results, and if those remain positive, pre-IND regulatory meetings with the UK MHRA and the US FDA CBER could begin.
In the medium term, roughly six months to two years out, the focus will shift to clinical translation preparation. The critical milestone is establishing GMP-grade manufacturing processes — upgrading the lab protocol to meet clinical standards for cell culture environments, bioreactor specifications, quality control metrics, and sterility procedures. This is where manufacturing costs will become clearer and the economic feasibility of the approach will be tested.
The first human clinical trial is expected to begin between late 2028 and 2029. The likely candidates will be children with long-gap oesophageal atresia who have failed conventional treatment or lack suitable surgical options. The trial design will probably be a safety-focused Phase I/II study with a small cohort of 5-10 patients. GOSH would be the natural first site, with Boston Children's Hospital or Children's Hospital of Philadelphia potentially joining as multi-center participants. During this period, convergence with 3D bioprinting technology could produce hybrid approaches — synthetic bioprinted scaffolds seeded with patient cells, eliminating dependence on animal-derived scaffolds entirely. Companies like Aspect Biosystems and BICO are already investing heavily in this direction.
Looking at the long-term horizon of two to five or more years, this research signals the birth of personalized organ medicine as a clinical discipline. In the bull case scenario, standardized culturing protocols for 3-5 tubular organs are established by the early 2030s, with regulatory approval in at least 10 countries. The tissue engineering market grows from $43.1 billion in 2030 to surpass $100 billion by 2035, with custom organs representing 15-20% of the total. The donor shortage for tubular organs is effectively resolved, and transplant waitlist mortality drops 30-40% from current levels.
The base case sees clinical approval for two organs — oesophagus and trachea — by the mid-2030s, available at 20-30 advanced medical institutions worldwide. Per-procedure costs settle around $100,000-250,000, initially higher than conventional reconstruction but cost-effective over time through reduced complication treatment. Insurance coverage remains limited, approved primarily for patients who have failed conventional treatment. Custom organs represent 5-10% of the tissue engineering market.
In the bear case, unforeseen complications in human trials delay development. If immune responses in humans are not fully controlled, or if long-term fibrosis or stricturing occurs, fundamental protocol revisions would be needed, potentially pushing regulatory approval beyond 2040. The technology would survive but clinical adoption would be significantly delayed. Even so, the tissue engineering data and cell culture advances accumulated during this process would benefit other regenerative medicine fields indirectly.
Three variables will determine which scenario unfolds. First, human cell engraftment rates on decellularized scaffolds — whether human muscle and progenitor cells attach and proliferate as efficiently as pig cells did. Second, the quality of nerve regeneration — the enteric nervous system controls peristalsis, and its complete reconstitution is essential for long-term swallowing function. Six months of nerve regeneration in pigs is encouraging but not a guarantee in humans. Third, the pace of manufacturing cost reduction — how quickly cell culture and bioreactor automation advances will determine whether this remains an elite procedure or becomes broadly accessible.
In the broader context of regenerative medicine, this study marks a pivotal milestone in the transition from medicine that repairs organs to medicine that manufactures them. Over 100,000 people worldwide are currently on organ transplant waiting lists, and in the United States alone, 17 people die every day waiting for an organ. Personalized cultured organs could fundamentally alter these tragic statistics. Complex solid organs like hearts and livers remain far more challenging to engineer than tubular structures, but success with tubular organs is the first step on that staircase.
Sources / References
- Engineered tissue offers hope for babies born with missing food pipe section — UCL News
- Lab-grown oesophagus restores pigs ability to swallow — Nature
- GOSH Charity-funded team engineers first lab-grown oesophagus — GOSH Charity
- Esophageal Atresia - Birth Defects — CDC
- Tissue Engineering Market Size To Reach $43.13Bn By 2030 — Grand View Research
- Engineered tissue offers hope for children born with missing esophagus — Medical Xpress
- Adult outcomes following neonatal oesophageal atresia — Nature Pediatric Research