A Lab-Grown Esophagus Survived Six Months Inside a Pig — The Story of the World's First Artificial Organ That Swallowed Food Without Immunosuppression
Summary
A GOSH-UCL team has achieved the world's first lab-grown esophagus transplant in pigs, restoring normal swallowing function for six months without immunosuppression. This breakthrough opens an entirely new treatment pathway for children born with esophageal atresia who currently face a lifetime of invasive surgeries.
Key Points
World's First Decellularized-Recellularized Esophagus Transplant in Large Animal Model
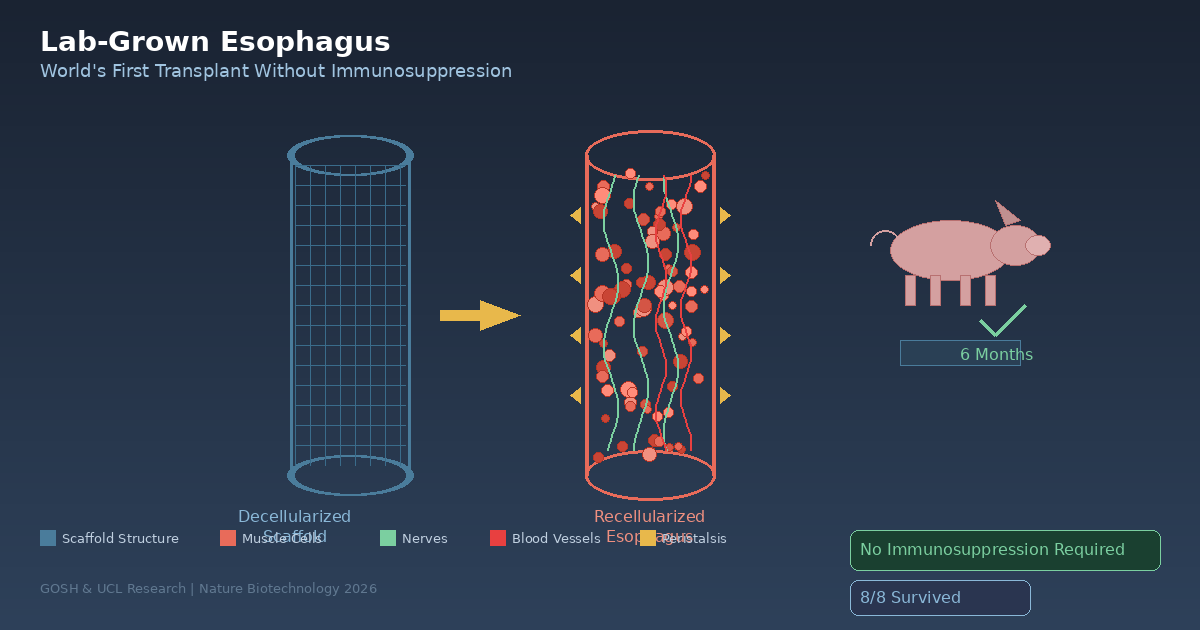
A research team from London's GOSH and UCL successfully transplanted lab-grown esophaguses into eight pigs after removing all cells from donor pig esophaguses and repopulating the scaffolds with recipient muscle cells. The two-month fabrication process yielded artificial esophaguses that fully integrated with surrounding tissue within three months and developed functional muscle, nerves, and blood vessels by six months, enabling peristalsis to move food normally. Most remarkably, all eight animals survived without any immunosuppressive drugs, experimentally proving that autologous cell-based approaches can bypass immune rejection — the greatest challenge in organ transplantation.
Revolutionary Treatment Pathway for Children with Esophageal Atresia
The ultimate target of this research is patients with esophageal atresia, a condition occurring in approximately 2.44 per 10,000 births where the esophagus is missing or severed. In long-gap cases, conventional surgery cannot bridge the gap, forcing radical alternatives using stomach or colon segments that leave 42.4% of patients with gastroesophageal reflux and 57.8% with dysphagia. An autologous cell-based artificial esophagus can grow with the child, reducing the need for repeat surgeries and restoring normal swallowing without immunosuppression — presenting a fundamentally different treatment paradigm.
The Valley of Death — Distance from Animal Studies to Human Application
Success in pigs does not automatically translate to human application. Fewer than 10% of promising preclinical technologies reach Phase 1 clinical trials, and the six-month observation window may be too short to detect chronic rejection that can manifest years after transplantation in humans. Differences between pig and human immune systems add another variable. The team's target of clinical trials within five years is aggressive by regenerative medicine standards, but their 2010 trachea transplant experience and GOSH's integrated research-clinical infrastructure provide support. Regulatory uncertainty, customized manufacturing costs, and the small sample size of eight animals remain key challenges.
Potential Paradigm Shift in Organ Donation — From Donation to Manufacturing
The long-term implications extend far beyond a single organ. The decellularization-recellularization technique is theoretically applicable to all tubular organs including bladders, ureters, blood vessels, and intestines. With 121,678 Americans currently on the transplant waiting list and 16 dying daily, and deceased donors declining 4% in 2025 (663 fewer transplants), autologous cell-based artificial organs herald a transition from dependency on others' deaths to manufacturing organs from patients' own cells. The global tissue engineering market projected to grow from $4.8 billion in 2024 to $9.8 billion by 2030 (12.8% CAGR) signals this transition is already economically materializing.
Medical and Economic Implications of Immunosuppression-Free Transplantation
Organ transplant recipients must take immunosuppressive drugs for life at costs reaching tens of thousands of dollars annually, while suffering severe side effects including increased infection vulnerability, elevated cancer risk, and kidney damage. If the autologous cell-based approach validated in this study demonstrates the same immune tolerance in humans, it could fundamentally improve transplant patients' quality of life while dramatically reducing long-term healthcare system costs. The impact is especially significant for pediatric patients who would be freed from decades of immunosuppressive medication.
Positive & Negative Analysis
Positive Aspects
- No Immunosuppression Required — A Game Changer for Transplant Medicine
The artificial esophagus built from the recipient's own cells is recognized as 'self' by the immune system, functioning without rejection. This sidesteps one of the greatest challenges in organ transplantation history. Freedom from immunosuppressive side effects (infections, cancer, kidney damage) enables normal life, and for pediatric patients specifically, liberation from decades of drug burden represents a revolutionary advance.
- Growing Artificial Organs — Meeting the Core Need of Pediatric Patients
Conventional transplanted organs cannot grow with a child, necessitating repeat surgeries. This study experimentally confirmed the theoretical basis that autologous cell-based grafts can grow alongside the recipient. The result showing pigs growing normally over six months while the artificial esophagus adapted supports a core hypothesis in pediatric regenerative medicine.
- Peristalsis Restoration — Beyond Structure to Function
Previous tissue engineering attempts mostly achieved structural replacement only. This study demonstrated nerve and blood vessel regeneration enabling active esophageal contraction and peristalsis. This constitutes decisive evidence that artificial organs can transition from inert structural substitutes to living, functional organs.
- Multi-Organ Expansion Potential — Beyond the Esophagus
The decellularization-recellularization platform is a versatile technology applicable to various tubular organs including bladders, ureters, blood vessels, and small intestines. A single success story can accelerate expansion to other organs, potentially addressing the global health crisis of organ shortage at its root.
- GOSH-UCL Integrated Research-Clinical Infrastructure
The ability to perform the entire pipeline from cell biopsy through lab culture to transplant surgery within a single institution accelerates the research-to-clinical transition. The 2010 experience with the world's first tissue-engineered trachea transplant will enhance regulatory confidence in esophagus clinical trials.
Concerns
- The Valley of Death Between Animal and Human Translation
The reality that fewer than 10% of preclinically successful technologies reach Phase 1 clinical trials cannot be ignored. Pig and human immune systems differ in response patterns, and the six-month observation period may be insufficient to capture chronic rejection that can manifest years post-transplant in humans. The fundamental limitation of life sciences — that successful animal experiments do not guarantee human success — must not be forgotten.
- Manufacturing Scalability and Cost Barriers
The process requiring cell harvest and two months of custom cultivation per patient makes mass production impossible. While precise cost estimates are unavailable, costs higher than conventional surgery are expected. The paradox that regenerative medicine's personalized nature could become a barrier to universal access is real, particularly problematic for children in developing countries with limited medical resources.
- Sample Size and Statistical Robustness Concerns
While eight pigs suffice for proof of concept, the sample is statistically weak for establishing safety and efficacy. Rare complications or long-term side effects may not be captured at this scale. Larger animal studies will almost certainly be required before human trials, entailing additional time, resources, and animal ethics discussions.
- Regulatory Framework Uncertainty
Customized decellularized grafts occupy a unique category fitting neither drug, medical device, nor biological product classifications precisely. Predicting which approval pathway regulatory agencies will apply is difficult, adding significant uncertainty to clinical trial design and timelines. If regulatory innovation fails to keep pace with technological innovation, situations may arise where technically ready treatments cannot reach patients.
Outlook
In the short term, over the next one to six months, this Nature Biotechnology paper will send significant ripples through the regenerative medicine community. Follow-up studies attempting to apply similar decellularization-recellularization techniques to other organs are likely to emerge rapidly, particularly in fields involving tubular structures such as bronchi, ureters, and blood vessels. The GOSH-UCL team will begin pre-regulatory discussions and intensify in vitro experiments using human cells.
In the medium term, between six months and two years, multi-center collaborative studies will likely be organized to verify the reproducibility of these results. Whether other institutions can independently replicate the GOSH-UCL protocol will be the critical variable determining this technology's credibility. Simultaneously, manufacturing process standardization and quality control frameworks must be developed. Ethics committee approvals for clinical trial design will also begin during this period. Investment capital in the tissue engineering sector is likely to concentrate on decellularization platform companies, potentially driving rapid valuation increases for related startups.
In the long term, looking at a two-to-five-year horizon, De Coppi's team's target of clinical trials within five years enters the realm of possibility. In the bull case, a lab-grown esophagus is transplanted into the first human patient within three years, and successful results could trigger a paradigm shift across all of regenerative medicine. In the base case, small-scale Phase 1/2 clinical trials begin around the five-year mark, confirming safety and initial efficacy. In the bear case, unexpected immunological complications or functional failures in human application force a retreat to additional basic research.
Looking further ahead, the success of this technology could redefine the very concept of organ donation. The transition from a system dependent on someone else's death to one where patients' own cells are used to manufacture the organs they need represents nothing less than a civilizational shift in medicine. With more than 121,000 people on the U.S. transplant waiting list alone and thousands dying each year while waiting, the urgency of this technological transition is self-evident. The projection that the tissue engineering market will grow to $9.8 billion by 2030 signals that this transition has already begun.
The GOSH-UCL lab-grown esophagus stands at the very front of this transformation. The immune tolerance demonstrated by the decellularization technique has the potential to fundamentally upend the paradigm of transplant medicine. In conventional organ transplantation, immunosuppressive drugs have been a necessary evil, but the autologous cell-based approach opens a path to eliminating that necessary evil altogether.
The growth of the tissue engineering industry can signify democratization of medical access beyond mere market expansion. Currently, organ transplantation is subject to countless variables including donor-recipient matching, waiting times, and geographic accessibility. If autologous cell-based artificial organs are commercialized, all these variables disappear. Organs can be manufactured from patients' cells, where patients are, for whatever patients need.
Of course, four gates must be passed before this future becomes reality: technological maturity, regulatory approval, cost reduction, and manufacturing standardization. Technologically, restoration of peristalsis in human cells must be confirmed to work identically to animals. Regulatorily, new approval pathways for customized biological products must be established. Cost-wise, the currently labor-intensive manufacturing process must be automated to reduce per-patient costs. For manufacturing standardization, protocols must be established enabling any hospital worldwide to produce artificial organs of identical quality.
If any one of these four gates fails, commercialization will be delayed, but if all four are breached, the history of medicine will be divided into the era of donation and the era of manufacturing. The GOSH-UCL team's artificial esophagus is the first concrete piece of evidence for this historic transition.
Sources / References
- Engineered tissue offers hope for children born with missing esophagus — Medical Xpress
- GOSH Charity-funded team engineers first lab-grown oesophagus in breakthrough for children's surgery — Great Ormond Street Hospital
- Engineered Esophagus Rebuilds Missing Organ Segment in Pig Models — Genetic Engineering & Biotechnology News
- Lab-Grown Esophagus Offers Hope for Children with Long-Gap Esophageal Atresia — RegMedNet
- Organ Donation Statistics — U.S. Department of Health & Human Services
- Transplant success for world's first functioning tissue-engineered oesophagus — National Institute for Health and Care Research
- Tissue Engineering Market Size To Reach $43.13Bn By 2030 — Grand View Research