Astrocytes Were Never Just 'Support Cells' — They Were the Master Switch of Fear Memory All Along
Summary
A groundbreaking 2026 Nature study has revealed that astrocytes in the basolateral amygdala actively encode, retrieve, and extinguish fear memories, shattering the century-old dogma that neurons alone govern memory. This discovery, combined with emerging astrocyte-targeting drug candidates like KDS2010 and corroborating findings on astrocyte engrams, signals a genuine paradigm shift in neuroscience with profound implications for PTSD treatment. With approximately 3.9% of the global population experiencing PTSD in their lifetime and current first-line treatments failing roughly 40% of patients, the astrocyte pathway opens an entirely new therapeutic frontier — but also raises urgent ethical questions about memory manipulation, military applications, and the boundary between healing and erasure.
Key Points
The End of the Neuron Monopoly — Astrocytes Rewritten as Memory Architects
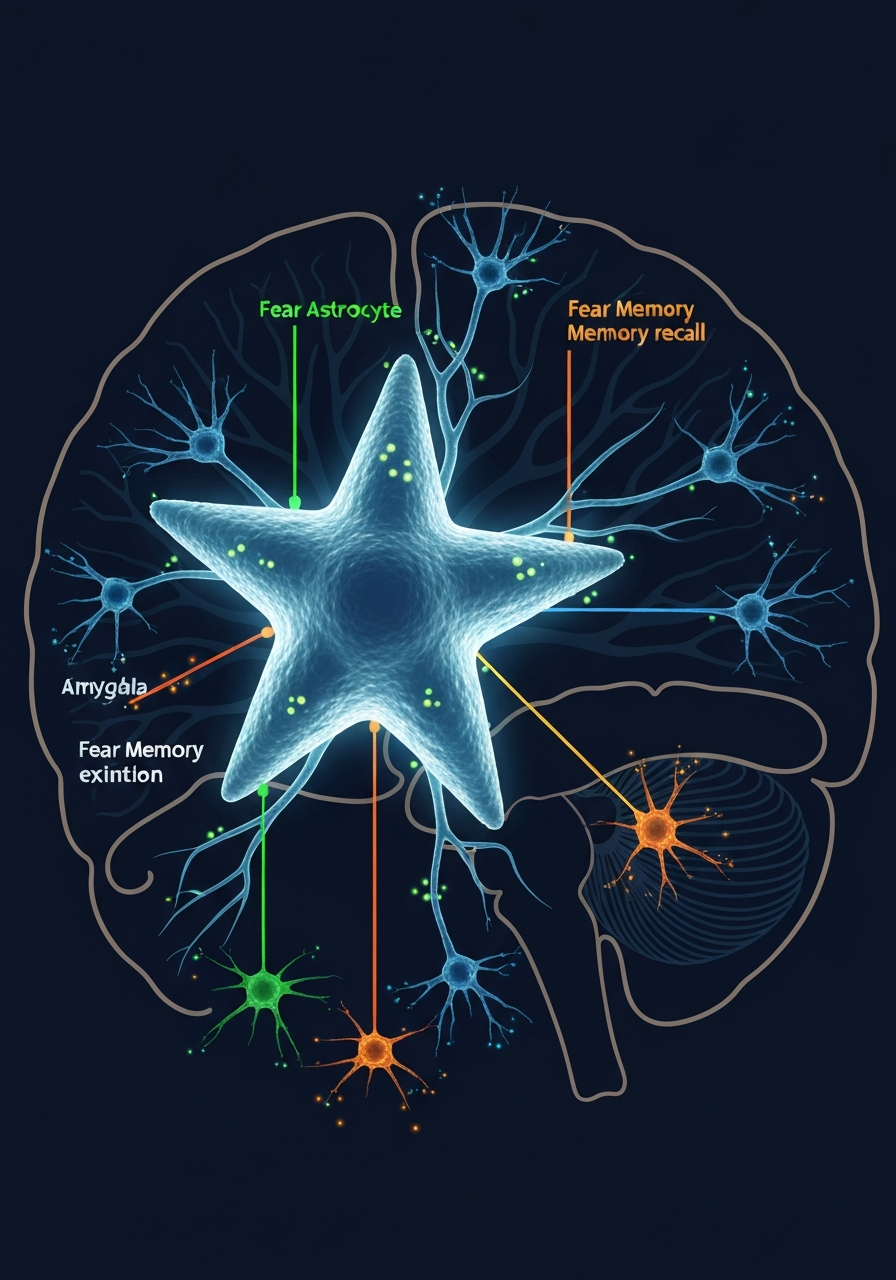
For more than a century, neuroscience operated under an unquestioned assumption: neurons do the thinking, and everything else is support staff. Astrocytes, the star-shaped glial cells that make up nearly half of the brain's roughly 170 billion cells, were dismissed as housekeeping units — maintaining ion balance, cleaning up neurotransmitters, feeding nutrients to the real workers. The February 2026 Nature paper from Andrew Holmes at the NIH Laboratory of Behavioral and Genomic Neuroscience and Lindsay Halladay at the University of Arizona has demolished that assumption with extraordinary precision. Using in vivo calcium imaging and chemogenetic manipulation in mouse models, their team demonstrated that astrocytes in the basolateral amygdala dynamically track fear states and actively enable neural fear signaling. As Halladay herself noted, 'For the first time, we found that astrocytes encode and maintain neural fear signaling.' This is not a minor refinement of existing theory. It is a fundamental rewriting of how we understand the cellular basis of memory, and it arrives alongside corroborating evidence from Nature 2024 and 2025 papers showing that astrocytes form their own learning-associated ensembles — dubbed 'astroengrams' — that stabilize memories over multiple days.
The Triple Switch — How Astrocytes Control Fear Memory Formation, Recall, and Extinction
What makes this discovery so consequential is not simply that astrocytes participate in fear memory. It is that they appear to control the entire lifecycle of fear memory through a three-phase mechanism that neuroscience had attributed exclusively to neuronal circuits. During fear learning, astrocyte calcium activity in the BLA increases markedly, facilitating the encoding of fear associations. During recall, these same astrocytes reactivate, supporting the retrieval of fear memories. During extinction — the process by which the brain learns that a previously threatening stimulus is no longer dangerous — astrocyte activity decreases, enabling the neural rewiring necessary for fear reduction. Critically, when researchers experimentally enhanced astrocyte signaling, fear memories strengthened. When they suppressed it, fear responses diminished. When they disrupted astrocyte activity entirely, the normal neuronal patterns associated with fear processing were impaired not just locally in the amygdala, but across the broader BLA-to-medial prefrontal cortex circuit. A comprehensive 2026 PMC review has since mapped astrocyte involvement across the entire amygdala-hippocampus-prefrontal cortex network, confirming that these cells play a fundamental role in fear memory acquisition, consolidation, extinction, and retrieval across distributed brain regions.
A New Horizon for PTSD Treatment — The Case for Astrocyte-Targeting Therapeutics
The clinical significance of the astrocyte discovery cannot be separated from the staggering burden of PTSD. According to the WHO, approximately 3.9% of the global population will experience PTSD in their lifetime, with about 354 million adult war survivors carrying PTSD or major depression. Among conflict-exposed populations, the prevalence reaches 15.3% — more than three times the general rate. In the United States alone, VA data shows that 29% of Iraq and Afghanistan veterans have experienced PTSD, with FY2024 figures showing 14% of male and 24% of female veteran service users diagnosed with the condition. Current first-line pharmacotherapy relies primarily on SSRIs, but a 2022 Cochrane meta-analysis of 1,078 patients found that only 58% showed symptom improvement compared to 35% on placebo, leaving roughly 40% of patients without adequate relief. Previous attempts at memory manipulation — notably propranolol-based reconsolidation blocking — have shown no significant overall effect on PTSD symptoms. Against this backdrop, the astrocyte pathway represents something genuinely new: a cellular target that governs fear memory at a level upstream of the neuronal circuits that current drugs attempt to modulate. KDS2010, or Tisolagiline, a reversible selective MAO-B inhibitor with an IC50 of 7.6 nM and 12,500-fold selectivity over MAO-A, is currently in Phase 2 trials for Alzheimer's disease, and its mechanism of suppressing astrocyte-mediated GABA production provides a proof-of-concept that astrocyte-targeting pharmacology is both feasible and clinically testable.
The Ethics of Fear Erasure — Identity, Autonomy, and the Limits of Healing
The possibility of modulating fear memories through astrocyte manipulation raises ethical questions that the scientific community cannot afford to defer. The AMA Journal of Ethics has identified at least six critical dimensions of concern surrounding memory manipulation: effects on learning, moral behavior, social harm prevention, privacy, commercialization, and conflicts of interest. At the heart of these debates is a deceptively simple question — if traumatic memories are constitutive of personal identity, does erasing them alter who a person fundamentally is? Neuroethicists have argued that traumatic memories, however painful, serve as integral components of narrative identity, informing moral reasoning, empathy, and social awareness. Survivors of atrocities have historically channeled their traumatic experiences into advocacy, social movements, and art that has transformed societies. A further concern raised in the academic literature is the risk that pharmaceutical companies could expand the clinical definition of 'trauma' to enlarge their market, pathologizing normal stress responses and creating demand for interventions that may not be medically necessary. The tension between individual autonomy — a patient's right to seek relief from unbearable suffering — and broader social considerations will only intensify as astrocyte-targeting therapies move closer to clinical reality.
The Military Temptation — When Fear Modulation Meets National Security
DARPA's investment history makes it clear that the US defense establishment views fear memory manipulation as a strategic capability, not merely a medical one. The SUBNETS program invested $70 million over five years to develop wireless deep brain stimulators for treating PTSD, major depression, and chronic pain in military personnel. The RAM program targets memory restoration after brain injury. Perhaps most revealing is the N3 program, which aims to develop nonsurgical bidirectional brain-computer interfaces specifically for 'able-bodied warfighters' — not patients, but active military operators. The NIH BRAIN Initiative, despite being cut from $680 million in FY2023 to $321 million in FY2025, remains a substantial research pipeline with an FY2026 budget request of $429 million. When astrocyte-targeting tools are added to this existing infrastructure of military neuroscience research, the dual-use implications become impossible to ignore. The same technology that could free a sexual assault survivor from paralyzing flashbacks could theoretically be used to suppress fear responses in combat soldiers, creating operators who function without the protective mechanism of fear. The line between therapeutic application and performance enhancement is not just blurry — it may not exist at all in institutional contexts where military necessity is the governing principle.
A Paradigm Shift Built on Converging Evidence, Not a Single Study
It is worth emphasizing that the Nature 2026 paper does not stand alone. The paradigm shift in our understanding of astrocytes rests on a convergence of multiple high-impact findings that have accumulated over the past two years. A 2024 Nature study demonstrated that learning induces c-Fos expression in specific hippocampal astrocytes, forming learning-associated astrocyte ensembles that directly contribute to memory recall — silencing the Fos gene in these astrocytes impaired both fear conditioning learning and recall. A 2025 Nature study went further, showing that astrocyte ensembles act as multiday traces that stabilize memories over time, introducing the concept of 'astroengrams' as a parallel to the neuronal engram concept that has dominated memory research for decades. A comprehensive PMC review has systematically documented astrocyte involvement in fear memory processing across the entire amygdala-hippocampus-prefrontal cortex network. Additionally, a PMC review on astrocytes in neurological disease has catalogued the expanding recognition that astrocyte dysfunction contributes to Alzheimer's, Parkinson's, ALS, and epilepsy, positioning these cells as therapeutic targets across a broad spectrum of conditions. This is not a single provocative finding — it is an emerging consensus, and the speed at which corroborating evidence is accumulating suggests that the neuron-centric paradigm's days are genuinely numbered.
Positive & Negative Analysis
Positive Aspects
- An Entirely New Therapeutic Target for Treatment-Resistant PTSD
The astrocyte pathway offers what decades of neuronal-focused research have not: a fundamentally different cellular target for fear memory disorders. With approximately 40% of PTSD patients showing inadequate response to SSRIs — the current gold standard — and propranolol-based memory reconsolidation approaches failing to demonstrate significant overall efficacy, the field has been running out of mechanistic ideas within the neuronal framework. Astrocyte-targeting therapeutics operate upstream of neuronal circuits, potentially modulating fear memory at a more fundamental level. KDS2010's Phase 2 status for Alzheimer's demonstrates that astrocyte pharmacology is already clinically viable, and redirecting similar approaches toward PTSD could open an entirely new treatment category for the estimated 354 million war survivors and millions of civilian PTSD sufferers worldwide.
- Converging Multi-Study Evidence Provides Exceptional Scientific Robustness
Unlike many paradigm-shift claims that rest on a single dramatic finding, the astrocyte revolution in memory science is built on independent corroborating evidence across multiple labs, methodologies, and brain regions. The 2024 Nature paper on learning-associated astrocyte ensembles, the 2025 Nature paper on astrocyte memory traces, and the 2026 Nature paper on fear memory encoding collectively form a coherent narrative that is extremely difficult to dismiss as artifact or coincidence. This convergence dramatically reduces the risk that the findings are domain-specific or methodologically flawed.
- Bidirectional Control Opens Precision Therapeutic Possibilities
The most therapeutically exciting aspect of the discovery is that astrocyte manipulation produces bidirectional effects on fear memory — enhancing signaling strengthens fear memories, while suppressing it diminishes them. This bidirectional control is precisely what clinicians need for nuanced therapeutic applications. Rather than blunt pharmacological suppression of entire neural circuits, astrocyte-targeting approaches could theoretically enable selective modulation of specific fear memories, potentially preserving adaptive fear responses while reducing pathological ones.
- Massive Market Opportunity Could Accelerate Drug Development
The PTSD treatment market, valued at approximately $2.24 billion in 2024 and projected to reach $2.85 billion by 2030, represents only the immediate addressable opportunity. The broader CNS therapeutics market stands at roughly $107 billion in 2024, projected to reach $156.5 to $209.2 billion by 2030, with mental health commanding a 34.13% market share. Astrocyte-targeting therapeutics that demonstrate efficacy in PTSD could be extended to anxiety disorders, phobias, and trauma-related conditions, substantially expanding the addressable market.
- Integration with Existing Neurostimulation Technologies
Understanding the astrocyte pathway does not only create opportunities for pharmacological intervention. It also has the potential to optimize existing non-pharmacological approaches such as transcranial magnetic stimulation and transcranial direct current stimulation. If astrocyte activity patterns can be mapped with sufficient precision, these stimulation technologies could be calibrated to target astrocyte-rich regions with parameters tuned to their specific calcium signaling dynamics.
Concerns
- The Mouse-to-Human Translation Gap Remains a Critical Unknown
All of the key findings — the 2024, 2025, and 2026 Nature papers — are based on mouse models. While mouse neuroscience has been an indispensable tool for understanding brain mechanisms, the history of translational neuroscience is littered with promising mouse findings that failed to replicate in humans. The human brain contains approximately 86 billion neurons and 84 billion glial cells, operating in a vastly more complex architecture than the mouse brain. Until human studies confirm that BLA astrocytes play a comparable role in human fear memory, the clinical applications remain speculative.
- Profound Ethical Risks of Memory Manipulation Technology
The ability to modulate fear memories raises ethical concerns that are not merely theoretical but have concrete implications for how this technology could be misused. If traumatic memories can be selectively weakened through astrocyte manipulation, the question of who decides which memories to modify becomes urgent. Neuroethicists have warned that traumatic memory is a constitutive element of personal identity, and its modification could alter personality, moral reasoning, and social bonds in unpredictable ways.
- Military Dual-Use Potential Creates Genuine Security Concerns
DARPA's existing investments in neurotechnology — $70 million for SUBNETS, the RAM memory restoration program, and the N3 nonsurgical brain-computer interface program explicitly designed for active warfighters — demonstrate that the US defense establishment views memory and fear modulation as strategic capabilities. The prospect of soldiers with pharmacologically suppressed fear responses is not science fiction in this context — it is a logical extension of programs already funded and underway.
- Research Funding Uncertainty Threatens the Pipeline
The NIH BRAIN Initiative budget has been cut from $680 million in FY2023 to $321 million in FY2025 — a reduction of more than 50%. While the FY2026 budget request of $429 million includes $195 million from the 21st Century Cures Act, that funding stream is set to expire after FY2026. This means that the fundamental research pipeline feeding astrocyte neuroscience faces genuine funding uncertainty at precisely the moment when the discoveries are most promising.
- Specificity and Off-Target Effects Remain Unresolved
Astrocytes are not confined to the amygdala — they are distributed throughout the entire brain and perform critical homeostatic functions including ion balance regulation, metabolic support, synaptic modulation, and neuroinflammation control. Any pharmacological intervention targeting astrocyte signaling pathways risks affecting these essential functions in brain regions unrelated to fear memory. KDS2010's mechanism of MAO-B inhibition is systemic, not anatomically targeted, meaning that its effects on astrocyte GABA production would occur brain-wide.
Outlook
There is a particular kind of scientific discovery that does not merely add a data point to existing knowledge but forces an entire field to re-examine its foundational assumptions. The February 2026 Nature paper on astrocytes and fear memory belongs to this category. For more than a century, neuroscience built its understanding of memory on a single premise — that neurons are the sole architects of learning, encoding, and recall, while glial cells exist merely to support the neurons' work. We now have converging evidence from three independent Nature publications in as many years that this premise was fundamentally incomplete.
I want to be precise about what this means and what it does not mean. This is not the death of neuronal memory theory. Neurons remain essential to memory formation and retrieval. What the astrocyte research reveals is that the brain's memory architecture is a dual system, not a single one — neurons and astrocytes working in concert through mechanisms we are only beginning to map. The concept of 'astroengrams,' introduced in the 2025 Nature paper, suggests that astrocytes maintain their own parallel memory traces that stabilize and support neuronal engrams over multiple days. This is a profoundly different picture from anything that appeared in neuroscience textbooks even three years ago.
In the short-term window of one to six months, the research community will focus on replication and extension. The Holmes-Halladay team's findings will be presented, debated, and tested by labs worldwide. Expect a surge of publications examining astrocyte calcium dynamics in other brain regions and memory types beyond fear conditioning. The critical question during this period is whether the BLA astrocyte findings generalize to the hippocampus for declarative memory, to the prefrontal cortex for working memory, and to the nucleus accumbens for reward-associated memory. If they do, the paradigm shift accelerates dramatically. If the effect proves specific to fear circuits in the amygdala, the impact narrows to PTSD and anxiety disorders — still enormously valuable, but contained.
During this same window, the pharmaceutical landscape will begin responding. KDS2010's ongoing Phase 2 trial for Alzheimer's disease, enrolling 114 patients in Korea with a US cohort planned for 2025, provides the closest existing proof-of-concept for astrocyte-targeting pharmacology. Its mechanism — selective MAO-B inhibition that suppresses astrocyte-mediated GABA production with an IC50 of 7.6 nM and 12,500-fold selectivity over MAO-A — is directly relevant to the fear memory findings, since GABA signaling in the BLA is central to the encoding and extinction of fear. If KDS2010's Alzheimer's data show cognitive benefits, pharmaceutical companies will rapidly explore whether the same compound or analogues could be repurposed for PTSD. Drug repurposing can compress development timelines from the typical 12 to 15 years for a novel molecule down to 3 to 5 years, because safety and pharmacokinetic profiles are already established.
The PTSD treatment market provides substantial commercial motivation for this pivot. Valued at approximately $2.24 billion in 2024 and projected to reach $2.85 billion by 2030 at a 4.1% CAGR according to Grand View Research, the narrowly defined PTSD therapeutics market is significant but understates the true opportunity. The broader CNS therapeutics market stands at roughly $107 billion in 2024 and is projected to reach between $156.5 billion and $209.2 billion by 2030, with mental health commanding a 34.13% market share. An astrocyte-targeting drug that demonstrates efficacy in PTSD would have obvious extension pathways into generalized anxiety disorder, specific phobias, and trauma-related conditions — potentially addressable markets an order of magnitude larger than PTSD alone.
For the medium-term outlook spanning six months to three years, the critical variable is the mouse-to-human translation question. All three of the landmark astrocyte papers rely on mouse models, and the history of translational neuroscience demands caution. The human brain's 86 billion neurons and 84 billion glial cells operate in an architecture of vastly greater complexity than the mouse brain. Astrocyte subtypes, regional distribution, and signaling dynamics may differ meaningfully between species. The propranolol experience provides an instructive parallel — strong preclinical evidence for memory reconsolidation blocking in rodents failed to translate into consistent clinical PTSD outcomes, with a meta-analysis showing no significant overall effect despite individual studies showing promise in severe cases.
However, several factors suggest the astrocyte story may translate more successfully than propranolol did. First, the astrocyte mechanism targets a cellular population rather than a single molecular pathway, providing a broader and potentially more robust intervention point. Second, modern neuroimaging technologies — particularly advances in fMRI resolution and PET ligands specific to glial activity — are approaching the capability needed to directly observe astrocyte dynamics in the living human brain, something that was impossible even five years ago. If human neuroimaging studies confirm that BLA astrocyte activity patterns correlate with fear memory in the same way they do in mice, the translational case strengthens enormously. Third, the convergence of three independent lines of evidence — fear encoding in 2026, memory recall ensembles in 2024, and multiday memory stabilization in 2025 — creates a coherent mechanistic narrative that is harder to dismiss than isolated findings.
During this medium-term window, the ethical and regulatory landscape will begin crystallizing. The AMA Journal of Ethics has already identified six critical dimensions of concern surrounding memory manipulation: effects on learning capacity, moral behavior, social harm prevention, privacy, commercial exploitation, and institutional conflicts of interest. The central tension — between a patient's autonomy to seek relief from unbearable suffering and the broader social implications of memory modification — will intensify as clinical applications approach feasibility. Neuroethicists have argued that traumatic memories, however devastating, are constitutive elements of personal identity, and their modification or erasure could alter personality, moral reasoning, and interpersonal bonds in ways that are difficult to predict and impossible to reverse.
The pharmaceutical commercialization risk also deserves serious attention during this period. Academic literature has flagged the danger that the clinical definition of 'trauma' could be expanded by pharmaceutical companies seeking to enlarge their addressable market, effectively pathologizing normal stress responses and creating demand for interventions that are not medically indicated. The opioid epidemic provides a grim precedent for what happens when pain management becomes a commercial growth opportunity divorced from clinical necessity. Robust regulatory frameworks specifically governing memory-modifying therapeutics need to be developed proactively, before the drugs reach market, not reactively after commercial pressures have already shaped prescribing patterns.
The military dimension will also become increasingly visible during this period. DARPA's existing portfolio — SUBNETS at $70 million over five years for wireless deep brain stimulation targeting PTSD and depression, the RAM program for brain injury memory restoration, and the N3 program developing nonsurgical brain-computer interfaces explicitly for 'able-bodied warfighters' — establishes that the US defense establishment views fear and memory modulation as strategic capabilities. The astrocyte discovery adds a potentially more accessible pharmacological pathway to this existing infrastructure. The VA's FY2024 data showing that 29% of Iraq and Afghanistan veterans have experienced PTSD, with overall rates of 14% for male and 24% of female veteran service users, ensures that there is both institutional motivation and a massive patient population driving military interest in astrocyte-based interventions.
The absence of international treaties governing cognitive enhancement or memory modification in military contexts is a genuine gap that could lead to an unregulated race. Unlike biological weapons or nuclear technology, cognitive neuroscience tools occupy a gray zone where the distinction between therapeutic use and performance enhancement is genuinely ambiguous. A drug that helps a veteran overcome PTSD flashbacks is a therapeutic. The same drug administered before deployment to suppress fear responses in active combat soldiers is an enhancement — and potentially a human rights violation, depending on whether informed consent is meaningfully possible in a military command structure. These are not hypothetical scenarios. They are the logical extension of programs that are currently funded and operational.
The research funding landscape adds another layer of uncertainty. The NIH BRAIN Initiative's budget trajectory — from $680 million in FY2023 to $321 million in FY2025, with an FY2026 request of $429 million that includes $195 million in 21st Century Cures Act funding set to expire after FY2026 — means that the fundamental research pipeline is under pressure at precisely the moment when discoveries are most promising. If Cures Act funding sunsets without replacement, the basic science that feeds the astrocyte-to-clinical translation pipeline could stall, pushing timelines back by years and ceding ground to research institutions in China, Europe, and South Korea that are increasing their neuroscience investments.
For the long-term outlook of three to ten years, I see three distinct scenarios that bracket the range of plausible outcomes.
The bull scenario envisions rapid translation success. Human neuroimaging studies by 2027 to 2028 confirm that BLA astrocyte dynamics mirror the mouse findings. KDS2010 or an analogue enters Phase 2 trials specifically for PTSD by 2028 to 2029, with preliminary efficacy data by 2030. Astrocyte-targeting approaches are integrated into combination therapies alongside existing SSRIs and trauma-focused psychotherapy, improving overall treatment response rates from the current 58% to 75% or higher. The PTSD treatment market expands beyond its current $2.24 billion trajectory as new drug categories reach market. International regulatory frameworks for memory-modifying therapeutics are developed proactively, and the dual-use military risks are managed through multilateral agreements analogous to the Chemical Weapons Convention. In this scenario, the 2026 Nature paper is remembered as the moment that PTSD treatment fundamentally changed, comparable to the discovery of SSRIs in the 1980s.
The base scenario — which I consider most probable — involves gradual, domain-specific progress. Human studies confirm astrocyte involvement in fear memory by 2028 to 2029, but the mechanistic details prove more complex than mouse models suggest, requiring additional years of basic research before clinical applications are viable. Astrocyte-targeting drugs enter Phase 2 PTSD trials by 2030 to 2031, with results available by 2032 to 2033. During this period, the primary clinical impact comes not from new drugs but from the incorporation of astrocyte biology into existing treatment frameworks — better understanding of why some patients respond to SSRIs and others do not, optimization of neurostimulation protocols based on astrocyte dynamics, and improved biomarkers for treatment selection. The ethical and military concerns remain active areas of debate but do not produce binding international regulation before 2032. Astrocyte-based PTSD treatments become available in limited clinical settings by the mid-2030s, a full decade after the initial discovery.
The bear scenario must be honestly confronted. Human studies reveal that the mouse findings do not cleanly translate — astrocyte subtypes differ enough between species that the precise BLA mechanism identified in mice does not operate identically in humans. The specificity problem proves intractable: systemic astrocyte-targeting drugs produce unacceptable off-target effects because astrocytes throughout the brain are affected, and targeted delivery technologies remain insufficient. KDS2010's Alzheimer's trial produces disappointing efficacy results, dampening pharmaceutical investment in the astrocyte pathway. Research funding constraints, particularly the expiration of Cures Act money and continued BRAIN Initiative budget pressure, slow the basic science pipeline. In this scenario, the astrocyte discovery enriches fundamental neuroscience but does not produce clinical PTSD treatments within a ten-year horizon. The paradigm shift in understanding occurs, but the therapeutic revolution is deferred.
What I am watching most closely across all these scenarios is not any single clinical trial or research paper but the convergence rate of human evidence. If, within the next two to three years, human neuroimaging studies confirm astrocyte dynamics consistent with the mouse findings, the bull scenario becomes considerably more likely and pharmaceutical investment will accelerate accordingly. If that confirmation is delayed or equivocal, the timeline extends and the base scenario prevails.
There is also a broader implication of this research that extends well beyond PTSD. If astrocytes are genuine co-architects of memory — and the evidence increasingly suggests they are — then our understanding of Alzheimer's disease, epilepsy, traumatic brain injury, and even normal aging may need substantial revision. The PMC review cataloguing astrocyte dysfunction across neurological diseases positions these cells as therapeutic targets for conditions that collectively affect hundreds of millions of people worldwide. The 2026 fear memory paper may ultimately be remembered not for what it revealed about fear specifically, but for what it revealed about the brain's fundamental operating architecture — that the organ we thought we understood had been running a dual operating system all along, and we had only been studying half of it.
The stakes are not abstract. The WHO estimates that approximately 3.9% of the global population experiences PTSD in their lifetime. About 354 million adult war survivors carry PTSD or major depression. Conflict-exposed populations face a 15.3% prevalence rate, more than three times the general population. In the United States, 29% of Iraq and Afghanistan veterans have experienced PTSD. Every year that effective treatment is delayed represents continued suffering for millions of people whose brains are trapped in fear loops that current medicine cannot adequately break. The astrocyte discovery does not guarantee a cure. But for the first time in decades, it offers a genuinely new direction — not another variation on the neuronal theme, but an entirely different chapter of the story. Whether that chapter leads to liberation or to new forms of manipulation depends on decisions that scientists, regulators, and societies will make in the coming years. The science has given us a new tool. What we do with it is the question that matters most.
Sources / References
- Astrocytes Enable Amygdala Neural Representations Supporting Memory — Nature
- These Overlooked Brain Cells May Control Fear and PTSD — ScienceDaily
- Learning-Associated Astrocyte Ensembles Regulate Memory Recall — Nature
- The Astrocytic Ensemble Acts as a Multiday Trace to Stabilize Memory — Nature
- Astrocytes in Fear Memory Processing: Molecular Mechanisms Across the Amygdala-Hippocampus-Prefrontal Cortex Network — PMC / Frontiers in Neuroscience
- Scilex Bio Reports KDS2010 Phase 2 Trial for Alzheimer's Disease Currently Enrolling — BioSpace
- Newly Developed Reversible MAO-B Inhibitor Circumvents the Shortcomings of Irreversible Inhibitors in Alzheimer's Disease — Science Advances
- Post-Traumatic Stress Disorder Fact Sheet — World Health Organization
- How Common Is PTSD in Veterans? — US Department of Veterans Affairs
- Pharmacotherapy for Post-Traumatic Stress Disorder (PTSD) — Cochrane Library
- Manipulating Memories: Ethics of Yesterday's Science Fiction and Today's Reality — AMA Journal of Ethics
- Erasing Traumatic Memories: When Context and Social Interests Can Outweigh Personal Autonomy — PMC / Philosophy, Ethics, and Humanities in Medicine
- DARPA SUBNETS Program — BRAIN Initiative Alliance
- Post-Traumatic Stress Disorder Treatment Market Report 2030 — Grand View Research
- Astrocyte in Neurological Disease: Pathogenesis and Therapy — PMC / Signal Transduction and Targeted Therapy