Water Was Never Just One Liquid — The Second Liquid Scientists Found at -63°C
Summary
After 34 years, the second critical point in supercooled water has been experimentally proven, reinterpreting the origin of water's 70-plus anomalies.
Key Points
The Experimental Full Stop on a Century of Water Theory
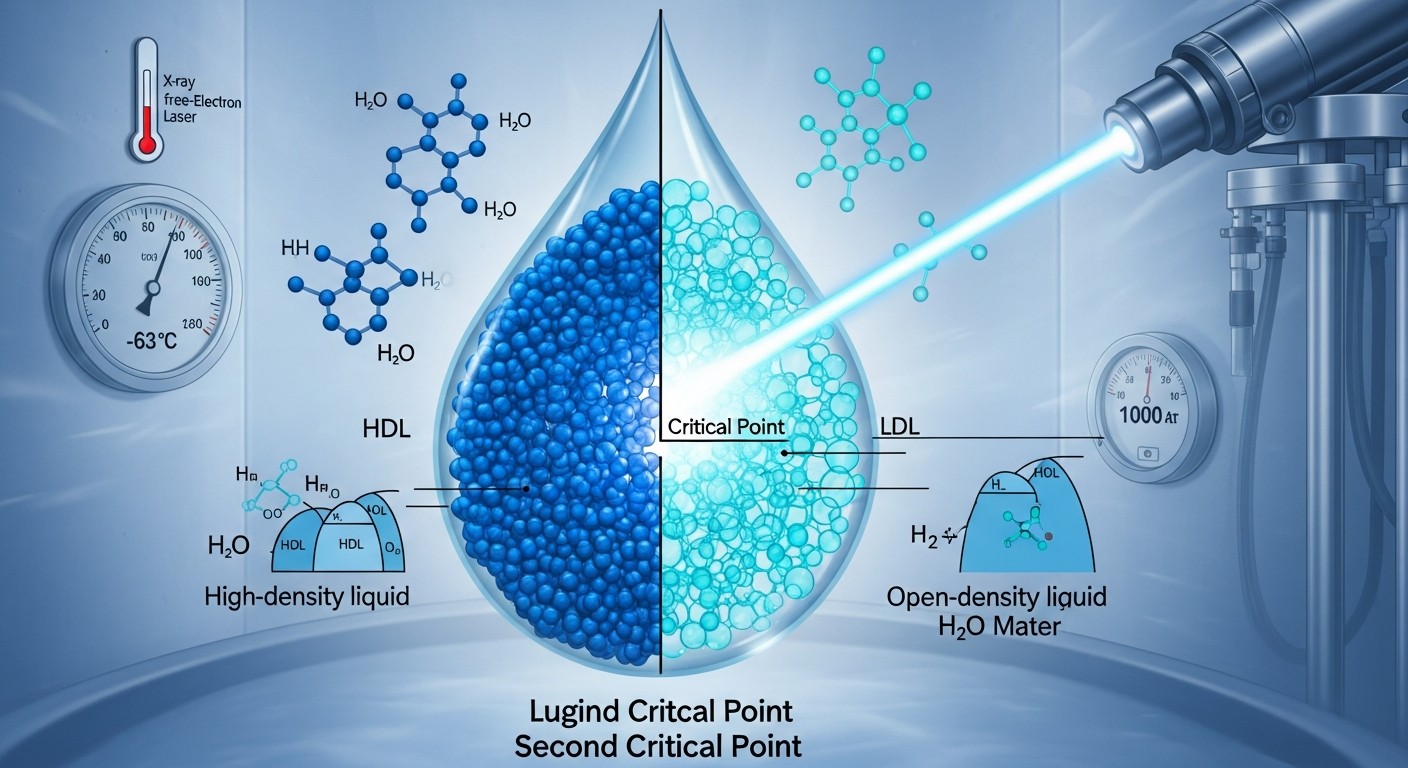
In 1992, Professor Eugene Stanley's team at Boston University used molecular dynamics simulations to predict a second critical point in water. Thirty-four years later, that prediction has been confirmed experimentally. Professor Anders Nilsson's team at Stockholm University captured the critical point where high-density liquid water (HDL) meets low-density liquid water (LDL) under extreme conditions of -63°C and 1,000 atmospheres, using ultrafast X-ray laser scattering. The results were published in Science, putting to rest one of the longest-running debates in water physics. Some researchers had insisted the simulation results were nothing more than computational artifacts, but this experiment has definitively overturned that claim. This stands as a landmark case where the predictive power of computer simulations was validated by experimental science, eloquently affirming the complementary relationship between theory and experiment.
A Unified Explanation for Water's 70-Plus Anomalies
Water is famously strange. It reaches maximum density at 4°C, expands when it freezes, and has an anomalously high heat capacity, among more than 70 documented anomalous behaviors. This discovery provides a unified explanation: all of these anomalies originate from enormous density fluctuations generated at the hidden critical point. Even at room temperature, water molecules are microscopically oscillating between high-density and low-density configurations, and the epicenter of those fluctuations is the second critical point at -63°C. What this means is that dozens of individually explained phenomena have now converged under a single underlying principle, which physicists are calling one of the most elegant unifications in the field. The fact that separate mechanisms previously invoked for each anomaly have collapsed into one common origin marks a genuine paradigm shift in our understanding of the most common substance on Earth.
Femtosecond X-ray Lasers — The Tool Revolution That Made the Impossible Possible
The Stockholm team developed an ingenious method: they flash-heated amorphous ice with an infrared laser and then captured the fleeting instant before the water recrystallized, using femtosecond-scale (one quadrillionth of a second) X-ray scattering. This experiment was physically impossible without X-ray free-electron lasers (XFELs), cutting-edge facilities that represent billions of euros in scientific infrastructure investment across Europe, the United States, and Japan. There are currently approximately 8 XFEL facilities worldwide, and this breakthrough vindicates the enormous investment that built them. Beyond water research, femtosecond X-ray techniques are applicable to protein folding dynamics, chemical reaction intermediates, and nanomaterial structural analysis. This is a textbook example of how advances in experimental tools unlock entirely new frontiers of scientific discovery.
A Fundamental Connection to the Conditions for Life
The fact that water is the only supercritical liquid at room temperature still influenced by this hidden critical point is directly tied to the existence of life. Without water's anomalous properties, lakes would freeze from the bottom up rather than the top down, exterminating aquatic life. Without its unusually high heat capacity, Earth's climate-regulating ability would vanish, causing wild temperature swings across the planet's surface. This can be interpreted as a chemical version of the anthropic principle: just as the universe's physical constants appear fine-tuned for life, water's molecular structure may be optimized for biology. This finding also becomes a key clue for predicting water's behavior in the subsurface oceans of moons like Europa and Enceladus, potentially resetting the entire direction of the search for extraterrestrial life. The discovery raises a profound question about whether water's peculiarity is merely a chemical coincidence or something deeper.
From Cryopreservation to Exoplanets — A Multi-Layered Cascade of Practical Implications
The practical implications of this discovery span multiple domains. In cryopreservation, it provides critical knowledge for designing new vitrification strategies that prevent ice crystallization, establishing the theoretical foundation to dramatically extend organ preservation times currently limited to just 4–6 hours. In atmospheric science, more precise modeling of supercooled water droplet behavior in clouds is expected to reduce uncertainties in IPCC climate projections. In the food industry, it enables optimization of rapid-freezing technologies, while in pharmaceuticals it can improve lyophilization (freeze-drying) processes. NASA's Europa Clipper and ESA's JUICE missions will directly apply these findings to predict water behavior in the subsurface oceans of outer solar system moons. This is a vivid demonstration of how a single fundamental science discovery can simultaneously ripple across medicine, climate science, food technology, and space exploration, making a powerful case for the value of basic research investment.
Positive & Negative Analysis
Positive Aspects
- A Historic Victory for Basic Science and Validation of the Scientific Method
A theoretical prediction that resisted experimental verification for 34 years has finally been confirmed, dramatically vindicating science's capacity for self-correction and the value of patience. In an era when public trust in science is fraying, this delivers a powerful message that science ultimately finds answers. It stands as a defining case where computer simulation predictions were validated by experiment, reaffirming the complementary relationship between theory and observation. The Stockholm team's success represents the fruition of Europe's long-term investment in scientific infrastructure, providing compelling justification for sustained basic science funding.
- Revolutionary Potential in Cryopreservation Could Transform Transplant Medicine
Understanding water's two liquid states opens entirely new strategic avenues for vitrification technology. Current organ preservation windows are brutally short — roughly 4–6 hours for a heart — and approximately 668,000 people across 75 reporting countries are on organ transplant waiting lists (GODT 2024), with approximately 5,000 per year dying while waiting in the United States alone. If new pathways that prevent ice crystallization can be engineered, preservation times could be extended from hours to days or weeks, potentially saving thousands of lives. This brings us one step closer to realizing organ banking, one of the most urgent challenges in 21st-century medicine.
- Improved Precision in Climate Prediction Models Strengthens Policy-Making
The behavior of supercooled water droplets in clouds is the single largest source of uncertainty in climate models. According to IPCC AR6, cloud radiative forcing uncertainty is a core variable in climate sensitivity estimates, currently at 2.5–4.0°C with a best estimate of 3.0°C. This discovery could refine microphysical models of supercooled droplets sufficiently to narrow that uncertainty range by 0.3–0.5°C — a margin that makes a real difference in climate policy formulation and strengthens the credibility of carbon emission targets. With IPCC AR7 drafting set to begin in earnest during 2026–2027, the timing of this discovery is particularly significant.
- A New Framework for the Search for Extraterrestrial Life
The subsurface oceans of moons like Europa and Enceladus exist under extreme pressure conditions of hundreds to thousands of atmospheres. This discovery provides the essential theoretical foundation for predicting how water behaves under those conditions. NASA's Europa Clipper and ESA's JUICE missions are scheduled to arrive around 2030, and these findings will directly inform the interpretation of mission data. New hypotheses are already emerging that the liquid-liquid phase transition in water could hold the key to the origin of life in these environments, potentially reshaping the entire direction of astrobiology research.
- Broad Applicability of Ultrafast X-ray Laser Methodology
The femtosecond X-ray scattering technique deployed in this experiment is far from limited to water research. It serves as a versatile platform applicable to real-time observation of protein folding, capturing transition states of chemical reactions, and structural analysis of nanomaterials. The successful demonstration of this methodology will increase utilization of XFEL facilities worldwide — including LCLS in the United States, SACLA in Japan, and the European XFEL in Germany — and open the floodgates for new experimental proposals. It is a vivid testament to the principle that tool innovation drives scientific discovery.
Concerns
- The Chronic Underfunding of Basic Science Collides with Short-Termism
This discovery was only possible because Europe invested billions of euros in XFEL infrastructure, yet globally, basic science budgets are stagnating or shrinking. The U.S. NSF budget has seen near-zero real growth after inflation adjustment, and South Korea's 2026 basic science R&D budget actually decreased year-over-year. The 34-year timeline of this discovery illustrates how the inherent nature of basic research — long-term investment with uncertain outcomes — structurally conflicts with modern funding systems that demand short-term results. Ensuring that research like this remains possible requires a renewed societal consensus on the value of scientific investment.
- Extreme Experimental Conditions Create High Barriers to Independent Replication
There are approximately 8 facilities worldwide equipped with femtosecond X-ray free-electron lasers, and competition for beam time is fierce. This means the number of teams capable of performing independent replication experiments is extremely limited, which could slow verification of a core principle of science: reproducibility. Precisely replicating the conditions of -63°C at 1,000 atmospheres is itself a technical challenge, and subtle differences in experimental setup could potentially influence results. Full academic consensus may take several years to achieve.
- The Long Road to Practical Applications and the Expectation-Reality Gap
Translating a phenomenon discovered at -63°C and 1,000 atmospheres into everyday technology is an entirely separate challenge. Applications like cryopreservation breakthroughs or climate model refinement could take a minimum of 5–10 years, and potentially more than 20 years. During that interval, researchers face relentless demands to justify their work with a persistent 'so what is this actually good for?' In a cruel paradox, if media hype inflates expectations beyond what the science can deliver in the near term, the eventual gap between promise and reality could strengthen skepticism toward basic science as a whole.
- Risk of Pseudoscience Exploitation and Public Misunderstanding
The provocative shorthand 'water comes in two kinds' is ripe for misinterpretation by the general public. Products peddling pseudoscience — 'structured water,' 'hexagonal water,' and similar nonsense — already exist in the marketplace. This discovery is highly likely to be selectively quoted in pseudoscience marketing: 'even the scientists say there are two kinds of water.' Such distortions could fuel the promotion of unscientific health supplements and wellness products. Accurate science communication has never been more critical, and both researchers and media bear shared responsibility for getting this right.
- Structural Difficulties in Workforce Development and Expert Cultivation
Training specialists in the highly specialized intersection of water physics and extreme-condition experimentation takes an extraordinarily long time. A doctoral program alone requires 5–7 years, and gaining hands-on XFEL operational experience demands additional years on top of that. The global pool of qualified experts in this field is very thin, and it must compete for talent against fields like high-energy physics and life sciences that attract far more funding. While this discovery could serve as a catalyst to draw young researchers into the field, resolving the structural workforce shortage requires dedicated policy efforts that go well beyond a single headline-making publication.
Outlook
Let me start with what will happen in the next few months. Once this paper is officially published in Science, I expect the reaction from the scientific community to be explosive. The Stockholm team's experiment will trigger a flood of independent verification requests at the world's premier X-ray free-electron laser facilities — SACLA (SPring-8 Angstrom Compact Free Electron Laser) in Japan, LCLS (Linac Coherent Light Source) at Stanford, and the European XFEL in Hamburg. My estimate is that at least two or three independent replication papers will emerge by the second half of 2026. That would be remarkably fast by the standards of this field, which tells you just how intense the community's hunger for this result has been.
The molecular dynamics simulation community will be equally busy in the near term. Existing models that predicted water's two liquid states — ST2, TIP4P/2005, mW, and others — will immediately be benchmarked against the new experimental data, and I expect this process to push the precision of water molecule simulations to a new level. For years, theorists and experimentalists have debated whether these computational models were capturing something real or generating artifacts from their own approximations. Now there is a concrete experimental reference point. That matters enormously for the credibility of computational physics as a whole, not just for water science.
Meanwhile, as the news value of this discovery spills beyond the academic community into mainstream media, water physics — a relatively niche discipline — is likely to see a temporary spike in both public attention and funding. This is the kind of result that could land on the cover of Science or Nature, which means the next six months represent a golden window for grant applications in related fields. The critical question is whether this interest becomes anything more than a transient media buzz. For that to happen, follow-up research needs to come quickly, connecting the discovery to tangible applications across multiple domains. Without those connections, there is a real risk that it gets filed away as 'that interesting water thing' and the momentum dissipates. It is worth noting that Chemistry World has already published balanced reporting that includes skeptical perspectives from scientists who urge caution before declaring the debate fully settled, and Technology Networks has covered the experimental details extensively, underscoring both the magnitude of the achievement and the remaining open questions.
The six-month to two-year window is where things start getting genuinely exciting. In cryopreservation, as our understanding of water's two liquid states deepens, I believe entirely new vitrification strategies will begin to emerge. The biggest enemy in organ cryopreservation today is ice crystal formation. Ice crystals shred cell walls, making it extremely difficult to preserve organs intact. But if we can precisely control the pathway through which water transitions via the high-density liquid state into a vitrified glass, the calculus changes dramatically.
We move one step closer to the organ bank — one of the most desperately sought technologies in 21st-century medicine. According to the Global Observatory on Donation and Transplantation (GODT) 2024 report, approximately 668,000 people across 75 reporting countries are on organ transplant waiting lists, with approximately 5,000 deaths per year occurring among those waiting in the United States alone. Current organ preservation windows are brutally short: roughly 4–6 hours for a heart, and slightly longer for kidneys. If preservation windows can be extended from hours to days or even weeks, thousands of lives could be saved annually.
Recent breakthroughs in the cryopreservation field offer reasons for cautious optimism. A 2023 study published in Nature Communications demonstrated successful vitrification and transplantation of rat kidneys, proving that small mammalian organs can survive the freeze-thaw cycle when ice formation is prevented. Even more encouragingly, a 2025 Nature Communications paper reported a milestone achievement in scaling vitrification to 3-liter volumes — approaching the size of human organs. These results suggest that the engineering challenges, while formidable, are not insurmountable. With the theoretical understanding now provided by the Stockholm team's discovery of how water navigates between its two liquid states, the design space for cryoprotectant solutions and cooling protocols expands significantly.
Atmospheric science is another domain where significant changes are expected. The IPCC's Seventh Assessment Report (AR7) enters active drafting during 2026–2027, and there is a strong possibility that findings about supercooled water droplet behavior will be incorporated into cloud microphysics models. Cloud radiative forcing is currently the single largest source of uncertainty in climate models, and a substantial portion of that uncertainty stems from our incomplete understanding of how supercooled water droplets behave in the upper atmosphere. According to IPCC AR6 Working Group 1 Chapter 7, the equilibrium climate sensitivity range stands at 2.5–4.0°C with a best estimate of 3.0°C.
If this discovery is integrated into AR7, I estimate the uncertainty range could narrow by 0.3–0.5°C. That may sound modest, but it is a margin that makes a real, material difference in climate policy formulation — affecting everything from carbon budget calculations to emission reduction targets. When policymakers debate whether to set a 1.5°C or 2.0°C warming target, the precision of the underlying climate sensitivity estimate is what determines whether a given emissions pathway is classified as safe or dangerous. Narrowing that uncertainty band translates directly into more credible and defensible policy positions. In the food industry, this knowledge can be applied to optimizing rapid-freezing technologies, particularly for cell-damage-free frozen seafood and improving the quality of cultured meat products.
The pharmaceutical industry is another beneficiary worth highlighting. Lyophilization, or freeze-drying, is a multi-billion-dollar process used to stabilize vaccines, antibiotics, and biologic drugs. The process fundamentally depends on how water behaves during freezing, and understanding the liquid-liquid transition pathway could enable more efficient lyophilization cycles that reduce energy consumption while improving product stability. Given the scale of global vaccine production and the push toward decentralized manufacturing in the post-pandemic era, even incremental improvements in freeze-drying efficiency could translate into meaningful cost savings and better cold-chain logistics.
Looking further out to the two-to-five-year horizon is where the truly transformative scenarios come into play. NASA's Europa Clipper and ESA's JUICE (JUpiter ICy moons Explorer) missions are scheduled to arrive at Jupiter's moons Europa and Ganymede around 2030. The subsurface oceans on these moons contain water under extreme pressure and temperature conditions — hundreds to thousands of atmospheres — and this discovery directly informs our predictions of how that water behaves. Europa's subsurface ocean lies beneath an ice shell tens of kilometers thick, at depths of roughly 100–200 km, where pressures reach hundreds to thousands of atmospheres.
Hypotheses are already emerging that the liquid-liquid phase transition in water could be a key factor in the origins of life under such conditions. The idea is that the density fluctuations associated with the critical point could create localized microenvironments where chemical reactions proceed differently than in uniform bulk water, potentially catalyzing the formation of prebiotic molecules. This is the kind of finding that could redirect the entire search for extraterrestrial life, shifting the focus from simply detecting water to understanding what state that water is in and how its phase behavior might enable or constrain biochemistry.
Textbooks will also change, and this matters more than it might initially seem. Current university physical chemistry textbooks present water's phase diagram with three states — solid, liquid, gas — and a single critical point. Adding a second critical point means revising foundational diagrams in physical chemistry and thermodynamics textbooks across the world. When changes happen at the textbook level, the ripple effects propagate across chemistry, biology, earth science, and materials science, shaping how an entire generation of scientists thinks about the most fundamental substance in nature.
In nanotechnology, it is already known that water behaves differently at the nanometer scale compared to bulk conditions. Confined water in carbon nanotubes, graphene oxide membranes, and biological ion channels exhibits anomalous properties that could be directly connected to the two-liquid-state framework. Designing nanofluidic devices and nanoreactors that exploit the liquid-liquid transition could open up entirely new engineering possibilities in areas ranging from desalination to drug delivery.
Let me break this down into scenarios with specific probabilities. In the bull case, independent replication succeeds within two to three years and a laboratory-level breakthrough emerges in cryopreservation — something like successfully preserving and transplanting a small organ such as a rat kidney after weeks of cryostorage. Investment in related biotech startups surges, water physics suddenly becomes a hot field attracting top young talent, and climate model precision improvements advance rapidly enough to be incorporated into the IPCC's interim assessment around 2028. I put the probability of this scenario at roughly 20%.
In the base case, full academic verification is completed within five years and water's phase diagram is officially revised. Cryopreservation and climate modeling improvements remain at the basic research stage, but the scientific community converges on the assessment that this is Nobel Prize-caliber work. Related research funding increases by 30–50%, though commercial applications remain distant. The discovery is recognized in the physics community as one of the most important experimental confirmations of the decade. I assign a 55% probability here.
In the bear case, independent replication experiments reveal subtle discrepancies, extending the debate. The existence of the critical point itself is not refuted, but consensus on its precise location and characteristics is delayed by a decade or more. Practical applications barely advance, and the discovery remains confined to the realm of pure academia. Part of the challenge is that approximately 8 XFEL facilities exist worldwide, as confirmed by a Nature Photonics 2024 review, and competition for beam time is intense enough to slow the pace of verification. I estimate a 25% probability for this scenario.
Of course, my predictions could be wrong. If an unexpected breakthrough emerges — say, a method for observing water's two liquid states at ambient temperature and pressure — that would be a game-changer on an entirely different level. There is also the possibility that quantum computing, once applied to water molecule simulations, could reveal phenomena that classical simulations never predicted. IBM's and Google's quantum computers are expected to reach the 100–1,000 qubit range around 2027–2028, which could be an inflection point for computational chemistry. The ability to simulate water molecule interactions from first principles, without the approximations inherent in classical force fields, would represent a qualitative leap in our understanding.
Looking at secondary and tertiary spillover effects into adjacent industries, we can expect deeper understanding of ultrapure water behavior in semiconductor manufacturing, where sub-nanometer contaminant control depends critically on water's phase behavior at interfaces. Optimization of lyophilization processes in pharmaceuticals will benefit from a more accurate thermodynamic model of water during freezing. Quality improvements in rapid-frozen food products — particularly the emerging cultured meat sector, where cellular integrity during freezing is a make-or-break quality factor — represent another tangible commercial pathway.
The semiconductor connection deserves particular attention. As chip fabrication moves to sub-2nm process nodes, the behavior of ultrapure water used in cleaning and etching becomes increasingly critical. Water's properties at solid-liquid interfaces, influenced by the underlying two-state framework, affect everything from wafer contamination rates to yield. Any improvement in modeling these interactions could save the industry billions in defect reduction.
If I may offer one piece of advice to readers: pay close attention when water-related research papers appear in Science or Nature going forward. I am convinced that this field will be among the most dynamic areas of fundamental science over the next five years. The convergence of ultrafast X-ray technology, computational simulation, and the profound implications for fields as diverse as medicine, climate science, and astrobiology creates a rare moment where basic research has the potential to reshape multiple domains simultaneously. We are living through one of those inflection points where a single discovery rewrites the foundations, and the ripple effects will be felt for decades. Whether the full promise is realized quickly or slowly, the direction is clear: we are entering a new era in our understanding of the most familiar substance on Earth. Thirty-four years of patience have produced an answer, and the questions that answer unlocks will keep scientists busy for decades to come.
Sources / References
- X-ray Lasers Enable Discovery of Critical Point in Supercooled Water — ScienceDaily
- Experimental Observation of the Liquid-Liquid Critical Point in Supercooled Water — Science (AAAS)
- Water's Hidden Critical Point Has Finally Been Observed — Science News
- X-ray Lasers Enable Discovery of Critical Point in Supercooled Water — Phys.org
- Scientists Find Critical Point of Supercooled Water — Sci.News
- Supercooled Water's Second Critical Point: What the Skeptics Say — Chemistry World
- The Discovery of Water's Second Liquid State in Detail — Technology Networks
- Successful Vitrification and Transplantation of Rat Kidneys — Nature Communications (2023)
- Breakthrough in 3-Liter Scale Vitrification for Organ Preservation — Nature Communications (2025)
- IPCC AR6 WG1 Chapter 7: The Earth's Energy Budget, Climate Feedbacks and Climate Sensitivity — IPCC
- Global Observatory on Donation and Transplantation 2024 Report — GODT / WHO
- Global XFEL Facility Landscape and Scientific Output Review — Nature Photonics (2024)